 Back
BackStudy Guide: Periodic Table, Chemical Nomenclature, Reactions, and Mole Concept
Study Guide - Smart Notes

Chapter 5: The Periodic Table
Classification of Elements by Period and Group
The periodic table organizes elements by periods (rows) and groups (columns), which helps predict their properties and behaviors. Elements in the same group share similar chemical properties.
Periods: Horizontal rows; elements in the same period have the same number of electron shells.
Groups: Vertical columns; elements in the same group have similar valence electron configurations.
Families: Notable groups include Alkali Metals (Group 1), Alkaline Earth Metals (Group 2), Halogens (Group 17), and Noble Gases (Group 18).
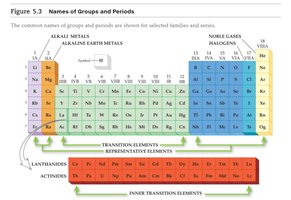
Trends in Metallic Character
Metallic character increases down a group and decreases across a period from left to right. Metals are typically found on the left and center of the periodic table.
Metallic character: Tendency to lose electrons and form positive ions.
Trend: Increases down a group, decreases across a period.
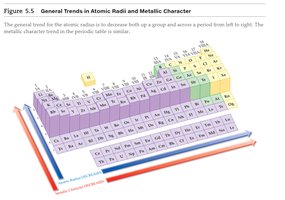
Trends in Atomic Size
Atomic radius decreases across a period and increases down a group due to changes in nuclear charge and electron shielding.
Atomic radius: Distance from nucleus to outermost electron shell.
Trend: Decreases left to right, increases top to bottom.
Predicting Physical and Chemical Properties
Elements in the same group often have similar physical and chemical properties, such as melting points, boiling points, and reactivity.
Example: Alkali metals are all highly reactive and have low melting points.
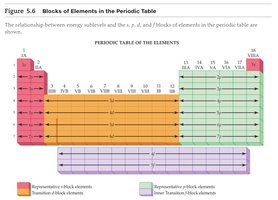
Electron Configuration and Sublevels
The electron configuration of an element can be predicted based on its position in the periodic table. The highest energy sublevel is determined by the block (s, p, d, f) in which the element resides.
Blocks: s-block, p-block, d-block, f-block.
Valence electrons: Number of electrons in the outermost shell; determines chemical reactivity.
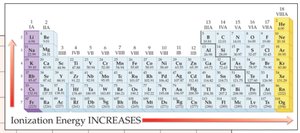
Ionization Energy Trends
Ionization energy is the energy required to remove an electron from an atom. It increases across a period and decreases down a group.
Highest ionization energy: Noble gases.
Lowest ionization energy: Alkali metals.
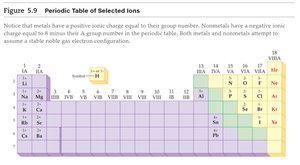
Periodic Table of Selected Ions
Metals typically form positive ions (cations), while nonmetals form negative ions (anions). The charge is often predictable based on group number.
Group 1: +1 charge
Group 2: +2 charge
Group 17: -1 charge
Chapter 6: Language of Chemistry
Classification of Compounds
Chemical compounds are classified based on their composition and bonding.
Binary ionic compounds: Composed of two elements, one metal and one nonmetal.
Ternary ionic compounds: Contain three or more elements, often including polyatomic ions.
Binary molecular (covalent) compounds: Composed of two nonmetals.
Classification of Acids
Binary acids: Contain hydrogen and one other element.
Ternary acids (oxyacids): Contain hydrogen, oxygen, and another element.
Classification of Ions
Monatomic cations: Single atom with positive charge (e.g., Na+).
Polyatomic cations: Multiple atoms with positive charge (e.g., NH4+).
Polyatomic anions: Multiple atoms with negative charge (e.g., SO42-).
Systematic Naming and Formulas
Monoatomic ions: Named based on element and charge (e.g., sodium ion, chloride ion).
Polyatomic ions: Named based on group (e.g., sulfate, nitrate).
Binary acids: Named as "hydro-" + root + "-ic acid" (e.g., hydrochloric acid).
Ternary acids: Named based on polyatomic ion (e.g., sulfuric acid).

Chapter 7: Chemical Reactions
Evidence of Chemical Reactions
Chemical reactions can be identified by observable changes.
Color change
Formation of a precipitate
Gas production
Temperature change
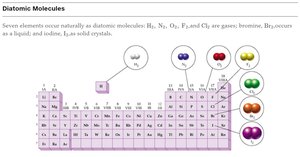
Diatomic Molecules
Seven elements naturally occur as diatomic molecules: H2, N2, O2, F2, Cl2, Br2, I2.
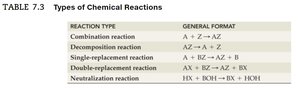
Types of Chemical Reactions
Chemical reactions are classified into five main types, each with a general format.
Reaction Type | General Format |
|---|---|
Combination | |
Decomposition | |
Single-replacement | |
Double-replacement | |
Neutralization |
Combination Reactions
Metal + oxygen: Forms metal oxide.
Nonmetal + oxygen: Forms nonmetal oxide.
Metal + nonmetal: Forms ionic compound.
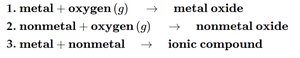
Decomposition Reactions
Metal hydrogen carbonate: Decomposes to metal carbonate, water, and carbon dioxide.
Metal carbonate: Decomposes to metal oxide and carbon dioxide.
Oxygen-containing compound: Decomposes to substance and oxygen gas.

Single-Replacement Reactions
Metal + salt: Forms new metal and salt.
Metal + acid: Forms salt and hydrogen gas.
Metal + water: Forms metal hydroxide and hydrogen gas.
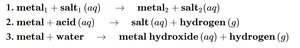
Activity Series Concept
The activity series ranks metals by their ability to undergo reactions. A metal higher in the series displaces a metal lower in the series. Highly active metals can displace hydrogen from water or acid.
Active metals: Li, Na, K, Ba, Sr, Ca
Displacement: Higher metal displaces lower metal or hydrogen.

Solubility Rules for Ionic Compounds
Solubility rules help predict whether an ionic compound will dissolve in water.
Rule | Soluble Compounds |
|---|---|
1 | Alkali metal ions, ammonium ion |
2 | Acetate ion, nitrate ion |
3 | Halide ions (except AgX, Hg2X2, PbX2) |
4 | Sulfate ion (except BaSO4, SrSO4, PbSO4) |
Rule | Insoluble Compounds |
5 | Carbonate, phosphate, sulfide, hydroxide ions (with exceptions) |

Chapter 8: The Mole Concept
Avogadro’s Number and the Mole
The mole is a fundamental unit in chemistry representing 6.02 x 1023 particles (atoms, molecules, ions). It allows chemists to count and measure substances at the atomic scale.
Avogadro’s number:
1 mole: Contains Avogadro’s number of particles.
Calculations Involving Moles, Mass, and Particles
Number of atoms in a molecule: Multiply the number of molecules by the number of atoms per molecule.
Mass of 1 mole: Use molar mass from the periodic table.
Calculating moles:
Calculating mass:
Molar Volume and Gas Calculations at STP
Molar volume at STP: per mole of gas
Density of gas:
Volume of gas:
Percent Composition and Empirical/Molecular Formulas
Percent composition:
Empirical formula: Simplest whole-number ratio of elements in a compound.
Molecular formula: Actual number of atoms in a molecule;

Additional info: All equations are provided in LaTeX format for clarity and academic rigor. Tables are recreated for comparison and classification purposes. Images are included only when directly relevant to the explanation of the adjacent paragraph.
