 Back
BackStudy Notes: Gases and Their Properties (Chapter 11, Introductory Chemistry)
Study Guide - Smart Notes

Gases
Kinetic Molecular Theory
The kinetic molecular theory is a model used to explain the behavior of gases. It provides a framework for understanding gas properties and predicts their behavior under many conditions, though it is not perfect and breaks down at low temperatures and high pressures.
Gas particles are in constant, random motion.
They do not attract or repel each other; collisions are elastic.
There is a lot of space between gas particles compared to their size.
The average kinetic energy of gas particles is proportional to the temperature in kelvin.
Properties explained by kinetic molecular theory:
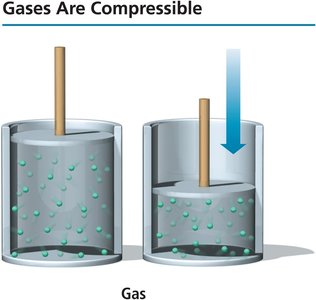
Compressibility: Gases can be compressed because there is much empty space between particles.
Shape and volume: Gases assume the shape and volume of their container.
Low density: Gases have much lower densities than liquids and solids.
Pressure
Pressure is the result of constant collisions between gas particles and the surfaces around them. It is defined as force per unit area:
Pressure formula:
Pressure depends on the number of gas particles in a given volume.
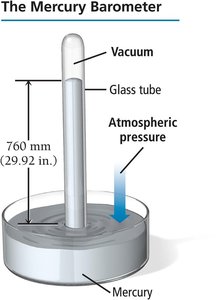
Units of Pressure
Pressure can be measured in several units:
Atmosphere (atm): Average pressure at sea level.
Pascal (Pa): SI unit,
Millimeter of mercury (mm Hg): Based on barometer measurements;
Torr:
Pounds per square inch (psi):
Inches of mercury (in. Hg):
Unit | Average Air Pressure at Sea Level |
|---|---|
Pascal (Pa) | 101,325 Pa |
Atmosphere (atm) | 1 atm |
Millimeter of mercury (mm Hg) | 760 mm Hg |
Torr | 760 torr |
Pounds per square inch (psi) | 14.7 psi |
Inches of mercury (in. Hg) | 29.92 in. Hg |
Pressure Unit Conversion
To convert between units, use the following relationships:
Example:
Gas Laws
Boyle’s Law (Pressure-Volume Relationship)
Boyle’s law states that the volume of a gas is inversely proportional to its pressure at constant temperature and amount:
Example: If pressure decreases from 4.0 atm to 1.0 atm, volume increases from 6.0 L to 24 L.
Charles’s Law (Volume-Temperature Relationship)
Charles’s law states that the volume of a gas is directly proportional to its temperature in kelvin at constant pressure and amount:
All temperatures must be in kelvin.
Combined Gas Law
The combined gas law relates pressure, volume, and temperature when the amount of gas is constant:
Avogadro’s Law (Volume-Amount Relationship)
Avogadro’s law states that the volume of a gas is directly proportional to the number of moles at constant temperature and pressure:
The Ideal Gas Law
The ideal gas law combines Boyle’s, Charles’s, and Avogadro’s laws into a single equation:
Where
P: pressure (atm), V: volume (L), n: moles, T: temperature (K)
Example: Calculate the volume occupied by 0.845 mol of nitrogen gas at 1.37 atm and 315 K:
Molar Mass of a Gas
The molar mass of a gas can be determined using the ideal gas law:
First, calculate moles:
Molar mass:
Example: If a sample has mass 0.136 g, volume 0.112 L, T = 298 K, P = 1.06 atm:
Relationships Between Gas Laws
Variable Quantities | Constant Quantities | Simple Gas Law |
|---|---|---|
V and P | n and T | Boyle's law: |
V and T | n and P | Charles's law: |
P and T | n and V | Gay-Lussac's law: |
P and n | V and T | |
V and n | T and P | Avogadro’s law: |
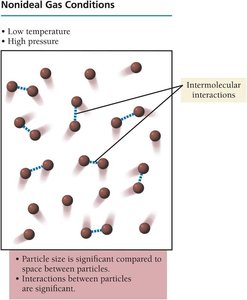
Ideal vs. Nonideal Gas Conditions
Gases behave ideally at high temperature and low pressure, where particle size is small compared to the space between particles and interactions are insignificant. At low temperature and high pressure, gases behave nonideally due to significant intermolecular interactions and particle size.

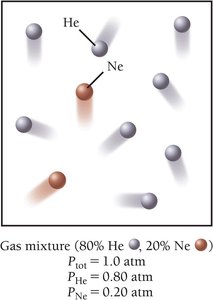
Partial Pressures: Mixtures of Gases
Each component in a gas mixture acts independently. The partial pressure of a component is its fractional composition times the total pressure:
Partial pressure = Fractional composition × Total pressure
Dalton’s law:
Example: In air, , ,
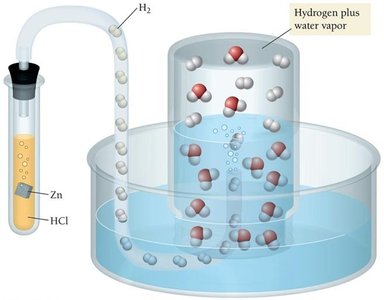
Vapor Pressure and Collecting Gases Over Water
When gases are collected over water, water vapor mixes with the gas. The pressure of water vapor is called vapor pressure and must be subtracted from the total pressure to find the pressure of the collected gas.
Example: If total pressure is 758 mm Hg and vapor pressure of water at 25°C is 23.8 mm Hg, then pressure of hydrogen gas is mm Hg.
Temperature (°C) | Vapor Pressure (mm Hg) |
|---|---|
10 | 9.2 |
20 | 17.5 |
25 | 23.8 |
30 | 31.8 |
40 | 55.3 |
50 | 92.5 |
60 | 149.4 |
70 | 233.7 |
80 | 355.1 |
Gases in Chemical Reactions
In reactions involving gases, the amount is often specified by volume at a given temperature and pressure. The ideal gas law is used to convert between pressure, volume, temperature, and moles, and stoichiometry is used to relate to other reactants or products.
Example: , then use stoichiometric coefficients to convert to other quantities.
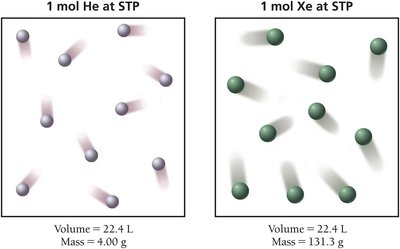
Molar Volume at Standard Temperature and Pressure (STP)
At STP (0°C, 1 atm), 1 mole of any ideal gas occupies 22.4 L. This is called the molar volume and is used as a conversion factor in calculations.
1 mole = 22.4 L at STP
Example: 0.879 mol CaCO3 produces 19.7 L CO2 at STP.
Example: How many grams of water form when 1.24 L of H2 gas at STP completely reacts with O2? Use molar volume and stoichiometry to solve.
