 Back
BackStudy Notes: Gases (Ch.11) – Introductory Chemistry
Study Guide - Smart Notes

Gases: Properties, Laws, and Applications
Pressure and Its Units
Pressure is a fundamental property of gases, defined as the force exerted per unit area by gas molecules colliding with the walls of their container. The SI unit for pressure is the Pascal (Pa), named after Blaise Pascal. Chemists also commonly use other units such as atmosphere (atm), millimeters of mercury (mmHg), and torr.
Pressure (P): Force per unit area.
Force (F): Measured in Newtons (N)
Area (A): Measured in square meters (m2)
Common Pressure Units:
Pascal (Pa)
Kilopascal (kPa)
Bar
Pounds per square inch (psi)
Atmosphere (atm)
Millimeters of mercury (mmHg)
Torr
Unit Conversions:
Example: If the same amount of gas is transferred from a 5.0 L container to a 10.0 L container, the pressure will decrease because the gas molecules have more space to move, resulting in fewer collisions per unit area.
Kinetic Molecular Theory (KMT)
The Kinetic Molecular Theory explains the behavior of gases by considering their molecular motion. It assumes gases are composed of tiny particles in constant, random motion, and provides a model for ideal gas behavior.
Postulate 1 – Volume: Gas particles are extremely small compared to the volume of their container.
Postulate 2 – Temperature: The average kinetic energy of gas particles increases with temperature.
Postulate 3 – Forces: Collisions between gas particles and container walls are perfectly elastic; there are no attractive or repulsive forces between particles.
Ideal Gas: An imaginary gas that perfectly follows the KMT assumptions.
Non-Ideal Behavior: At high pressures or low temperatures, real gases deviate from ideal behavior due to intermolecular forces and finite particle volume.
Example: At high pressure, the volume of gas molecules becomes significant, causing deviation from ideal behavior.
The Ideal Gas Law
The Ideal Gas Law relates the pressure, volume, temperature, and amount of gas in a sample. It is a central equation in gas chemistry.
Formula:
P: Pressure (atm or Pa)
V: Volume (L or m3)
n: Moles of gas
R: Gas constant ( or )
T: Temperature (K)
Example: To find the number of moles of NH3 in a 25.0 L tank at 190 ºC and 5.20 atm, use the Ideal Gas Law and convert temperature to Kelvin.
Derivations and Applications of the Ideal Gas Law
By rearranging the Ideal Gas Law, relationships between variables can be established, such as direct and inverse proportionality. These derivations are used when variables change between two states.
Direct Proportionality: If one variable increases, the other increases (e.g., V and T).
Inverse Proportionality: If one variable increases, the other decreases (e.g., P and V).
Derivation Example: (Combined Gas Law)
Example: If the number of moles inside a container is tripled while keeping pressure constant, the volume will triple.
Chemistry Gas Laws
Several gas laws describe the relationships between pressure, volume, temperature, and moles of gas under specific conditions.
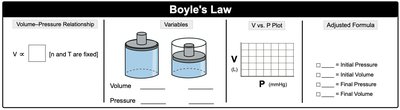
Boyle’s Law: (at constant n and T) – Volume and pressure are inversely proportional.
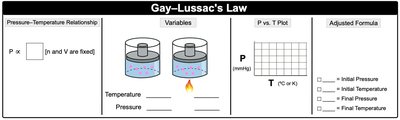
Gay-Lussac’s Law: (at constant n and V) – Pressure and temperature are directly proportional.
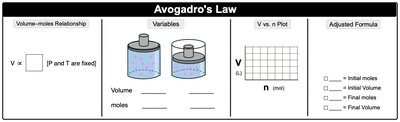
Avogadro’s Law: (at constant P and T) – Volume and moles are directly proportional.
Charles’s Law: (at constant n and P) – Volume and temperature are directly proportional.
Combined Gas Law
The Combined Gas Law merges Boyle’s, Charles’s, and Gay-Lussac’s Laws to relate pressure, volume, and temperature for a fixed amount of gas.
Formula:
Application: Used when more than one variable changes between two states.
Example: A gas initially at 900 mL, 520 K, and 1.85 atm changes to 330 mL and 770 K. The new pressure can be calculated using the Combined Gas Law.
Standard Temperature and Pressure (STP)
STP is a reference point for gas calculations. At STP, temperature is 0 ºC (273 K) and pressure is 1 atm. The molar volume of an ideal gas at STP is 22.4 L.
Standard Molar Volume: 1 mole of gas occupies 22.4 L at STP.
Conversion Factor: (at STP)
Example: To find the number of moles in 15.7 L of chlorine gas at STP, divide the volume by 22.4 L/mol.
Dalton’s Law of Partial Pressures
Dalton’s Law states that the total pressure of a mixture of gases is the sum of the partial pressures of each individual gas.
Formula:
Partial Pressure:
Fractional Composition:
Dalton’s Law (using fractional composition):


Example: If a gas mixture contains 72.8% chlorine and 27.2% neon by mass, and the total pressure is 809 mmHg, the partial pressure of neon is .
Gas Stoichiometry
Gas stoichiometry involves using balanced chemical equations to relate the volumes, masses, or moles of reactants and products in reactions involving gases.
Steps:
Convert the given quantity to moles.
Use the balanced equation to find the mole ratio.
Convert moles of unknown to desired units (volume, mass, etc.).
Example: To find the mass of Ag2O produced when 384 mL of O2 at 736 mmHg and 25 ºC reacts with excess Ag, use the Ideal Gas Law to find moles of O2, then use stoichiometry to find mass of Ag2O.
Additional info: All gas law calculations require temperature in Kelvin and pressure in consistent units. Real gases deviate from ideal behavior at high pressures and low temperatures.
