 Back
BackThe Mole and Chemical Formula Relationships: Study Notes for Introductory Chemistry
Study Guide - Smart Notes

The Mole
Definition and Importance
The concept of the mole is fundamental in chemistry, allowing scientists to count atoms, molecules, or ions in a given sample by relating mass to number of particles. The mole bridges the gap between the microscopic world of atoms and the macroscopic world of laboratory measurements.
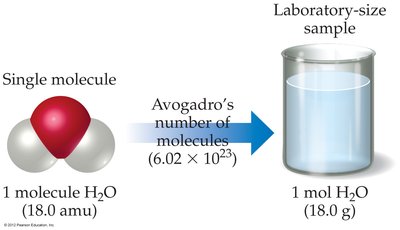
Mole: A unit representing 6.022 x 1023 particles (atoms, molecules, ions, etc.), known as Avogadro's Number.
Avogadro's Number: Defined as the number of particles in exactly 12 grams of carbon-12 (12C).
Application: Enables chemists to convert between mass and number of particles for any substance.



Avogadro's Number and Its Use
Avogadro's Number is experimentally determined and is used to relate the number of particles to the amount of substance.
1 mole = 6.022 x 1023 particles of anything.
For 12 grams of 12C, there are 6.022 x 1023 atoms.
Mole and Mass Relationship
The mass of one mole of a substance is called its molar mass, which is numerically equal to the sum of the atomic masses (in grams) of the elements in the compound.
Molar Mass: The mass (in grams) of one mole of a substance.
Examples:
1 mol O2(g) = 32.0 g
1 mol H2O(l) = 18.0 g
1 mol NaCl(s) = 58.45 g
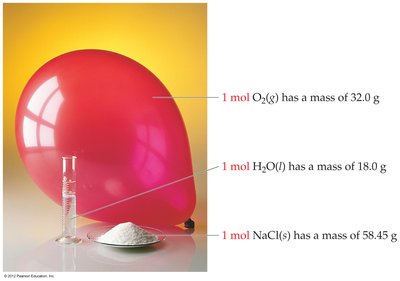
Calculating Moles and Mass
To convert between mass and moles, use the molar mass as a conversion factor.
Formula:
Example: How many moles are in 50.0 g of PbO2?
Molar mass of PbO2 = 207.2 (Pb) + 2 x 16.00 (O) = 239.2 g/mol
Chemical Formula Relationships
Mole Percent
Mole percent is the percentage of moles of one atom in a substance, calculated from the chemical formula.
Mole Percent:
Example: For C2H4O:
2 C moles, 4 H moles, 1 O mole (total = 7 moles)
C:
H:
O:

Mass Percent and Empirical Formula
Mass percent composition is the percentage by mass of each element in a compound. The empirical formula is the simplest whole-number ratio of atoms in a compound.
Mass Percent:
Empirical Formula: Determined from mass percent composition by converting masses to moles and finding the simplest ratio.
Example Table: Molar Masses of Common Substances
Substance | Molar Mass (g/mol) |
|---|---|
O2(g) | 32.0 |
H2O(l) | 18.0 |
NaCl(s) | 58.45 |
Summary
The mole is a central concept in chemistry, enabling quantitative analysis of chemical reactions and relationships. Understanding how to use Avogadro's Number, molar mass, and chemical formula relationships is essential for solving problems involving mass, moles, and percent composition. Key formulas:
Example applications:
Calculating the number of molecules in a sample
Determining the mass of a compound from the number of moles
Finding the empirical formula from mass percent composition
