 Back
Back1) The Octet Rule & Chemical Bonding: Introductory Chemistry Study Notes
Study Guide - Smart Notes

The Quantum Model of the Atom
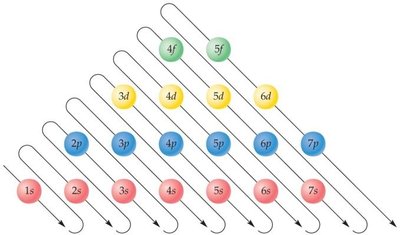
Main Shells, Subshells, and Orbitals
The quantum model describes the arrangement of electrons in atoms using shells, subshells, and orbitals. Electrons occupy energy levels (shells) around the nucleus, which are further divided into subshells (s, p, d, f) and orbitals.
Shells are labeled by principal quantum numbers (n = 1, 2, 3, ...).
Subshells are types of orbitals within each shell: s, p, d, f.
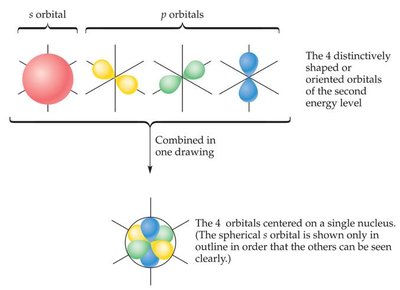
Orbitals are regions where electrons are likely to be found; each orbital can hold up to two electrons.
Electron configuration shows how electrons are distributed among shells and subshells.
Periodic Table and Electron Configuration
Using the Periodic Table to Determine Electron Configuration
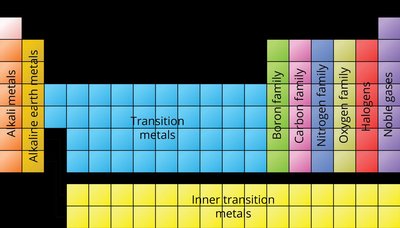
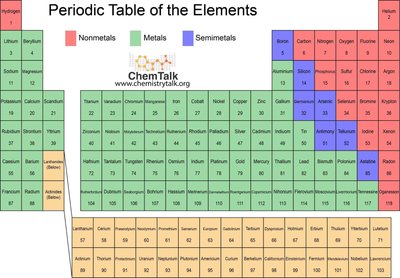
The periodic table is organized to reflect electron configurations. Elements are grouped into blocks (s, p, d, f) based on their outermost electrons.
s-block: Groups 1 and 2, plus Helium.
p-block: Groups 13-18.
d-block: Transition metals (Groups 3-12).
f-block: Inner transition metals (lanthanides and actinides).
Electron configuration notation (e.g., 1s22s22p5) shows the number of electrons in each subshell.
Valence Electrons and Chemical Bonding
Valence Electrons
Valence electrons are the electrons in the outermost shell of an atom. They determine chemical reactivity and bonding behavior.
For main group elements (Groups 1-20), the number of valence electrons corresponds to the group number.
Transition metals have more complex valence electron arrangements.
Valence electrons are crucial for understanding how atoms bond to form molecules and compounds.
Molar Mass and Molar Ratios
Calculating Molar Mass
Molar mass is the mass of one mole of a substance, expressed in grams per mole (g/mol). It is calculated by summing the atomic masses of all atoms in a chemical formula.
Example: The molar mass of CaMg(CO3)2 (dolomite) is calculated by adding the atomic masses of Ca, Mg, C, and O.
Percentage by mass of an element in a compound is found by dividing the mass of the element by the total molar mass and multiplying by 100%.
Stable Electron Configurations and the Octet Rule
The Octet Rule
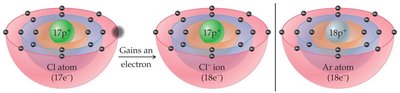
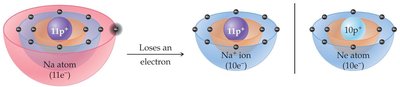
The octet rule states that atoms tend to gain, lose, or share electrons to achieve eight valence electrons, similar to the electron configuration of noble gases. This rule primarily applies to s and p block elements.
Noble gases are inert because they have full valence shells (eight electrons, except He).
Atoms become less reactive when their electron structure matches a noble gas.
Lewis Dot Structures
Visualizing Valence Electrons
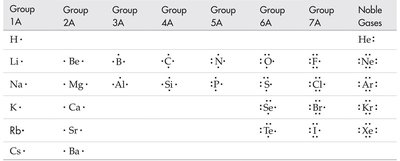
Lewis dot structures are diagrams that represent valence electrons as dots around the chemical symbol of an element. They help visualize bonding in molecules.
Each dot represents a valence electron.
Lewis structures are used for s and p block elements.
Practice: Draw Lewis dot structures for N, P, O.
Group 1A | Group 2A | Group 3A | Group 4A | Group 5A | Group 6A | Group 7A | Noble Gases |
|---|---|---|---|---|---|---|---|
H· | Be·· | B··· | C···· | N····· | O······ | F······· | Ne······· |
Li· | Mg·· | Al··· | Si···· | P····· | S······ | Cl······· | Ar······· |
Ionic and Covalent Bonds
Ionic Bonds
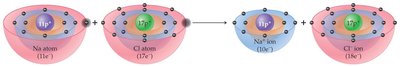
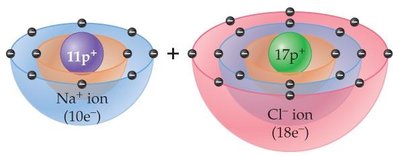
Ionic bonds form when atoms transfer electrons, resulting in the formation of positive (cations) and negative (anions) ions. The electrostatic attraction between oppositely charged ions holds the compound together.
Metals lose electrons to form cations (positive ions).
metals tend to lose an electron to reach the octet rule, By losing an electron, the form a cation anion, which is essentially a positive charged anion.
they are essentially too far away from the 8 valence electron, so to be closer they would rather lose and get to the + charge
Nonmetals gain electrons to form anions (negative ions).
nonmetals tend to gain electrons to reach the octet rule. By gaining electrons they are closer to the 8 valence electron. Also, by gaining electrons, they form negative anions.
Example: Sodium (Na) loses one electron to become Na+; chlorine (Cl) gains one electron to become Cl-.
Naming Ionic Compounds (a combination of both covalent and ionic bonds)
Ionic compounds are named by stating the cation first, followed by the anion. The cation name is the element name; the anion name is the root of the element plus the suffix '-ide'.
Example: NaCl is sodium chloride; MgO is magnesium oxide.
Transition metals may have multiple charges, indicated by Roman numerals (e.g., Fe2+ is iron(II)).
Ion | Name |
|---|---|
Na+ | Sodium ion |
Cl- | Chloride ion |
Fe2+ | Iron(II) ion |
Fe3+ | Iron(III) ion |
Cr6+ | Chromium(VI) ion |
Covalent Bonds
Covalent bonds form when two nonmetal atoms share one or more pairs of electrons. This sharing allows each atom to achieve a stable electron configuration.
can i say anions? two anions share one or more electrons?
Single, double, and triple covalent bonds involve sharing one, two, or three pairs of electrons, respectively.
Examples: Carbon forms covalent bonds in organic molecules; silicon forms covalent bonds in minerals.

Electronegativity and Bond Polarity
Electronegativity
Electronegativity is a measure of an atom's ability to attract electrons in a chemical bond. It influences the type of bond formed between atoms.
Higher electronegativity means stronger attraction for electrons.
Electronegativity increases across a period and decreases down a group.
Polar and Nonpolar Covalent Bonds
When atoms with different electronegativities form a bond, the electrons are unequally shared, resulting in a polar covalent bond. If the electronegativities are similar, the bond is nonpolar covalent.
Nonpolar covalent: Electronegativity difference < 0.5
Polar covalent: Electronegativity difference 0.5–2.0
Ionic: Electronegativity difference > 2.0
Example: H (2.1) and C (2.5) form a nonpolar covalent bond; H (2.1) and Cl (3.0) form a polar covalent bond.
Electronegativity Difference | Bonding Type |
|---|---|
< 0.5 | Nonpolar covalent |
0.5–2.0 | Polar covalent |
> 2.0 | Ionic |
Summary of Key Concepts
Molar mass and molar ratios are essential for quantitative chemistry.
Stable electron configurations and the octet rule explain chemical reactivity.
Ionic and covalent bonds are the two main types of chemical bonds.
Electronegativity determines bond polarity and type.
