 Back
BackThe Solvation Process: Dissociation and Hydration of Ionic Compounds
Study Guide - Smart Notes

The Solvation Process
Dissociation & Hydration of Ionic Compounds
The solvation process describes how ionic compounds, such as salt, dissolve in water. This process involves the physical separation of ions from their crystal lattice and their stabilization in solution by water molecules. Understanding solvation is fundamental to topics such as electrolytes, chemical reactions in solution, and biological transport.
Ionic Dissociation
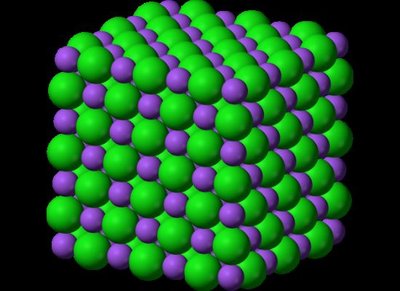
The Ionic Crystal Lattice
Before dissolving, ions in an ionic compound are locked in a rigid structure called a crystal lattice. This lattice is held together by strong electrostatic forces between positive cations and negative anions.
Electrostatic Forces: The attraction between oppositely charged ions keeps the lattice stable.
Stability: In the solid state, ions cannot move freely, so solid salt does not conduct electricity.
Energy Barrier: Breaking the lattice requires energy to overcome these attractive forces.
The Power of Polarity
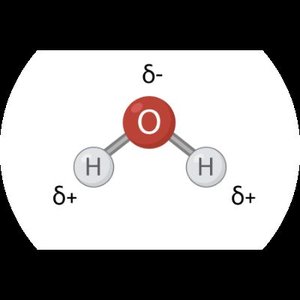
Water: The Polar Solvent
Water molecules are polar, meaning they have a separation of electric charge. This polarity is crucial for dissolving ionic compounds.
Oxygen End: Electronegative, carrying a partial negative charge (δ-).
Hydrogen Ends: Carry a partial positive charge (δ+).
When an ionic crystal meets water, the water molecules rotate to orient their charged ends toward the opposing ions. The partial negative charge of oxygen attracts cations, while the partial positive charge of hydrogen attracts anions.
Ion-Dipole Interactions
These interactions between water's dipole and the ions weaken the ionic bonds within the crystal, facilitating dissociation.
Water Oxygen (δ-): Attracts cations (+).
Water Hydrogen (δ+): Attracts anions (-).
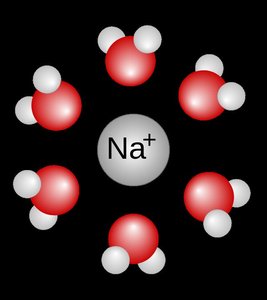
Formation of Hydration Shells
Hydration Shells and Stabilization
Once the attraction from water overcomes the lattice energy, ions are pulled into the solution. Each ion becomes surrounded by a sphere of water molecules, forming a hydration shell. This shell stabilizes the ion and prevents it from recombining with the crystal.
Hydration Shell: A cluster of water molecules surrounding a dissolved ion.
Stabilization: Hydration shells keep ions dissolved and mobile in solution.
Stepwise Solvation Process
Stages of Solvation
The solvation process can be broken down into four main steps:
Immersion: Ionic crystal is placed in water.
Orientation: Water aligns its partial charges (δ+ to anions, δ- to cations).
Dissociation: Forces overcome lattice bonds; ions separate.
Stabilization: Hydration shells form around free ions.
Applications and Importance
Why Solvation Matters
Solvation is not just about dissolving salt in water. It is a fundamental mechanism behind:
Electrolytes: Solutions that conduct electricity, essential for batteries and nerve signals.
Chemical Reactions: Free ions are more reactive than solid crystals, enabling complex chemistry in solution.
Biological Transport: How nutrients and minerals are transported through blood and cells.
Key Terms and Concepts
Solvation: The process of surrounding solute particles with solvent molecules.
Dissociation: The separation of ions from an ionic compound in solution.
Electrolyte: A substance that produces ions in solution and conducts electricity.
Hydration Shell: The cluster of water molecules surrounding a dissolved ion.
Ion-Dipole Interaction: The attraction between an ion and the partial charges of a polar molecule.
Relevant Equations
For sodium chloride (NaCl) dissociation in water:
General formula for ionic dissociation:
Table: Water-Ion Interaction Summary
Interaction Component | Charge | Attracts... | Target Ion |
|---|---|---|---|
Water Oxygen | Partial Negative (δ-) | Cations (+) | Na+, K+, etc. |
Water Hydrogen | Partial Positive (δ+) | Anions (-) | Cl-, SO42-, etc. |
Example: When table salt (NaCl) is added to water, it dissociates into Na+ and Cl- ions, each surrounded by hydration shells, creating an electrolyte solution.
Additional info: The solvation process is covered in detail in Chapter 4 of most introductory chemistry textbooks, under "Molecules and Compounds" and "Solutions." The images included visually reinforce the concepts of crystal lattice, water polarity, and hydration shells.
