 Back
BackUnit Conversion and Equalities in Chemistry
Study Guide - Smart Notes

Unit Conversion
Understanding Unit Conversion
Unit conversion is a fundamental skill in chemistry, allowing scientists and students to translate measurements between different systems of units. This process relies on equalities, which express the same quantity in two different units, and conversion factors, which are fractions derived from these equalities.
Equalities
Equalities use two different units to describe the same quantity. They can be between units of the metric system, U.S. system, or between metric and U.S. units.
Metric Equalities: 1 m = 1000 mm, 1 g = 1000 mg
U.S. Equalities: 1 lb = 16 oz, 1 ft = 12 in.
Metric–U.S. Equalities: 2.20 lb = 1 kg, 454 g = 1 lb
Example: Food labels often display both metric and U.S. units, indicating the same amount in two different units.

Equalities on Food Labels
Packaged foods in the United States frequently list contents in both U.S. and metric units. This practice helps consumers understand quantities regardless of their preferred measurement system.

Significant Figures in Equalities
Numbers in equalities between two metric units or two U.S. units are considered exact and do not affect significant figures. However, when an equality involves a metric and a U.S. unit, one number is measured and counts toward significant figures.
Exact Equalities: 1 g = 1000 mg, 1 ft = 12 in., 1 min = 60 s
Measured Equalities: 454 g = 1 lb, 946 mL = 1 qt, 39.4 in. = 1 m
Exception: 1 in. = 2.54 cm is defined as exact.
Common Equalities Table
The following table summarizes common equalities used in chemistry for length, volume, mass, and time.
Quantity | Metric (SI) | U.S. | Metric–U.S. |
|---|---|---|---|
Length | 1 km = 1000 m 1 m = 1000 mm 1 cm = 10 mm | 1 ft = 12 in. 1 yd = 3 ft 1 mi = 5280 ft | 2.54 cm = 1 in. (exact) 1 m = 39.4 in. 1 km = 0.621 mi |
Volume | 1 L = 1000 mL 1 dL = 100 mL 1 mL = 1 cm3 | 1 qt = 4 cups 1 gal = 4 qt | 946 mL = 1 qt 1 L = 1.06 qt 473 mL = 1 pt 5 mL = 1 tsp* |
Mass | 1 kg = 1000 g 1 g = 1000 mg* | 1 lb = 16 oz | 1 kg = 2.20 lb 454 g = 1 lb |
Time | 1 h = 60 min 1 min = 60 s | 1 h = 60 min 1 min = 60 s |
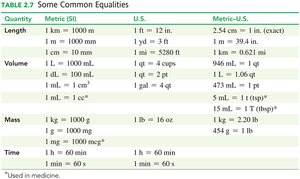
Additional info: Equalities marked with * are used in medicine.
Conversion Factors
Writing Conversion Factors
Any equality can be written as two conversion factors, which are fractions used to convert between units. Always include units in conversion factors.
For 1 h = 60 min: and
For 1 m = 1000 mm: and
Conversion Factors Within a Problem
Sometimes, equalities are stated within a specific problem and only apply to that context. For example, a car travels at 85 km/h, or a tablet contains 500 mg of vitamin C.
Equality | Conversion Factors | Significant Figures or Exact |
|---|---|---|
85 km = 1 h | 85 km/1 h and 1 h/85 km | 85 km is measured (2 SF); 1 h is exact |

Equality | Conversion Factors | Significant Figures or Exact |
|---|---|---|
1 tablet = 500 mg vitamin C | 500 mg vitamin C/1 tablet and 1 tablet/500 mg vitamin C | 500 mg is measured (1 SF); 1 tablet is exact |

Conversion Factors from a Percentage
A percent (%) can be used as a conversion factor by relating the parts of a unit to 100 parts of the whole. For example, 18% body fat by mass means 18 kg body fat per 100 kg body mass.
Equality | Conversion Factors | Significant Figures or Exact |
|---|---|---|
18 kg body fat = 100 kg body mass | 18 kg body fat/100 kg body mass and 100 kg body mass/18 kg body fat | 18 kg is measured (2 SF); 100 kg is exact |

Conversion Factors from Dosage Problems
Dosage problems in medicine often state equalities that can be written as conversion factors. For example, Keflex capsules contain 250 mg of cephalexin.
Equality | Conversion Factors | Significant Figures or Exact |
|---|---|---|
1 capsule = 250 mg Keflex | 250 mg Keflex/1 capsule and 1 capsule/250 mg Keflex | 250 mg is measured (2 SF); 1 capsule is exact |

Practice: Writing Equalities and Conversion Factors
Sample Problems
For each pair of units, write the equality and two conversion factors:
Liters and milliliters: 1 L = 1000 mL; and
Inches and meters: 1 m = 39.4 in.; and
Milligrams and micrograms: 1 mg = 1000 μg; and
Additional info: These conversion factors are essential for solving quantitative chemistry problems, such as calculating dosages, concentrations, and physical properties.
