 Back
BackBiomolecules and Their Role in Nutrition: Structure, Function, and Examples
Study Guide - Smart Notes

Organic Molecules and Functional Groups
Structure and Versatility of Carbon
Organic molecules are defined by the presence of both carbon (C) and hydrogen (H). Carbon's ability to form up to four covalent bonds, including double and triple bonds, allows for a wide variety of molecular structures, such as long chains, rings, and branched molecules. This versatility is foundational for the diversity of biomolecules essential to life.
Functional Groups
Functional groups are specific clusters of atoms attached to the carbon skeleton of organic molecules. They determine the chemical reactivity and polarity of the molecule, influencing its biological function.
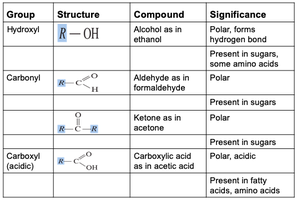
Group | Structure | Compound | Significance |
|---|---|---|---|
Hydroxyl | R-OH | Alcohol (ethanol) | Polar, forms hydrogen bonds |
Carbonyl | R-COH / R-CO-R | Aldehyde (formaldehyde), Ketone (acetone) | Polar, present in sugars |
Carboxyl (acidic) | R-COOH | Carboxylic acid (acetic acid) | Polar, acidic, present in fatty acids, amino acids |
Isomers and Enantiomers
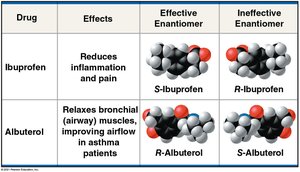
Isomers are molecules with identical molecular formulas but different atom arrangements. Enantiomers are a type of isomer that are mirror images due to the arrangement around an asymmetric carbon. Enantiomers can have different biological effects, especially in pharmaceuticals.
Drug | Effects | Effective Enantiomer | Ineffective Enantiomer |
|---|---|---|---|
Ibuprofen | Reduces inflammation and pain | S-Ibuprofen | R-Ibuprofen |
Albuterol | Relaxes airway muscles | R-Albuterol | S-Albuterol |
Biologically Important Organic Molecules
Categories of Biomolecules
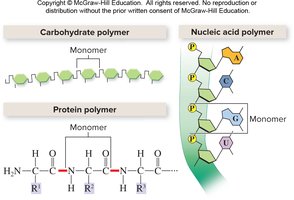
Biomolecules essential for life are classified into four main types: carbohydrates, proteins, nucleic acids, and lipids. Each category has distinct chemical structures, functions, and dietary sources.
Category | Subunits (Monomers) | Polymer |
|---|---|---|
Carbohydrates | Monosaccharide | Polysaccharide |
Lipids | Glycerol and fatty acids | Does not form polymers |
Proteins | Amino acids | Polypeptide |
Nucleic acids | Nucleotide | DNA, RNA |


Synthesis and Breakdown of Biomolecules
Monomers and Polymers
Biomolecules are often composed of repeating units called monomers. Polymers are formed by joining monomers together. Lipids are an exception, as they are not true polymers.
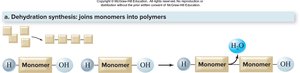
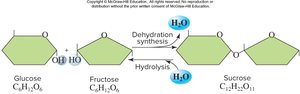
Dehydration Synthesis (Condensation Reaction)
Dehydration synthesis is the process by which enzymes join monomers to form polymers, releasing a water molecule. This reaction is essential for building complex biomolecules such as starch from glucose.
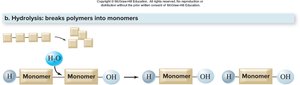
Hydrolysis
Hydrolysis is the process of breaking down polymers into monomers by adding a water molecule. This reaction is crucial for digestion, such as breaking down starch into glucose.
Enzymes: Catalysts of Chemical Reactions
Role and Mechanism
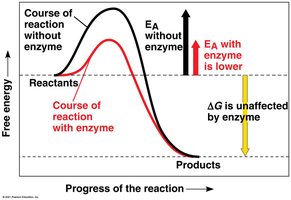
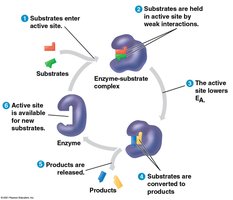
Enzymes are biological catalysts that speed up chemical reactions, including dehydration synthesis and hydrolysis. They are not consumed or altered by the reaction and are sensitive to pH and temperature.
Lower activation energy: Enzymes reduce the energy required for reactions.
Specificity: Each enzyme acts on specific substrates.
Carbohydrates
Structure and Function
Carbohydrates are composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. They serve as quick energy sources, structural components, and are involved in cell recognition. Carbohydrates are classified as monosaccharides, disaccharides, and polysaccharides.
Monosaccharides
Monosaccharides are simple sugars and the monomers of carbohydrates. They typically have 3 to 7 carbon atoms. Examples include glucose (blood sugar), fructose (fruit sugar), and galactose (found in avocados). Hexoses have six carbon atoms, while pentoses have five.

Disaccharides
Disaccharides consist of two monosaccharides joined by glycosidic linkages. Examples include lactose (galactose + glucose) and sucrose (glucose + fructose). Lactose intolerance results from the absence of the enzyme lactase.
Polysaccharides
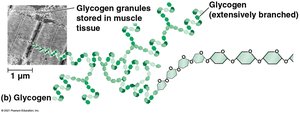
Polysaccharides are long chains of monosaccharides joined by glycosidic linkages. They serve as energy storage or structural molecules.
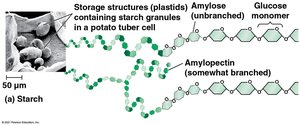
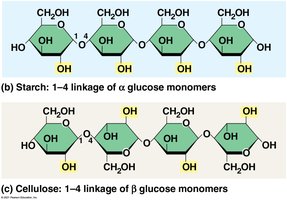
Starch: Energy storage in plants, digestible by humans.
Glycogen: Energy storage in animals, stored in liver and muscle, highly branched.
Cellulose: Structural component in plant cell walls, indigestible by humans but aids digestion.
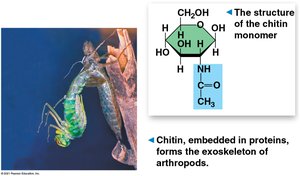
Chitin: Structural component in fungi and exoskeletons of arthropods.
Peptidoglycan: Structural component in bacterial cell walls.
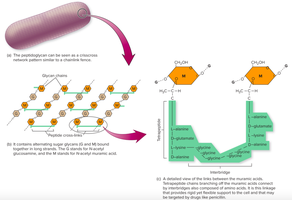
Lipids
Structure and Function
Lipids are composed of carbon, hydrogen, and oxygen, sometimes with additional elements. They are large, nonpolar, and hydrophobic molecules. Lipids serve as long-term energy storage, structural components, heat retention, cell communication, and protection. Types include fats, oils, phospholipids, steroids, and waxes.
Triglycerides
Triglycerides are fats and oils used for long-term energy storage. They consist of glycerol and three fatty acids.
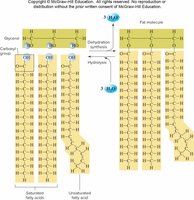
Types of Fats
Fatty acids can be saturated (no double bonds, solid at room temperature) or unsaturated (one or more double bonds, liquid at room temperature). Trans fats are unsaturated fats that have been hydrogenated to become solid.
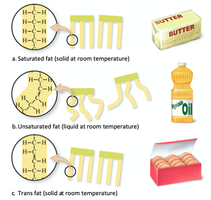
Proteins
Structure and Function
Proteins are polymers of amino acids linked by peptide bonds. They are composed of carbon, hydrogen, oxygen, nitrogen, and sometimes phosphorus. Proteins are essential for structure, function, and regulation in cells.
Primary structure: Sequence of amino acids.
Secondary structure: Alpha helices and beta sheets.
Tertiary structure: Three-dimensional folding.
Quaternary structure: Association of multiple polypeptides.
Amino Acids
There are 20 common amino acids, each differing by their R group. The sequence and properties of amino acids determine protein structure and function.
Nucleic Acids
Structure and Function
Nucleic acids are composed of carbon, hydrogen, oxygen, nitrogen, and phosphorus. Their monomer is the nucleotide, which consists of a phosphate group, a pentose sugar, and a nitrogenous base. Nucleic acids transmit genetic information and carry instructions for protein synthesis.
DNA: Stores genetic information.
RNA: Transfers genetic information and assists in protein synthesis.
ATP: Energy currency of the cell.
Nucleic Acid Bonding
Nucleotides are joined by phosphodiester bonds in DNA and RNA. Nitrogenous bases in DNA are held together by hydrogen bonds.
ATP: Adenosine Triphosphate
ATP is a nucleotide composed of adenine, ribose, and three phosphate groups. The last two phosphate bonds are unstable and easily broken, releasing energy for cellular work. Hydrolysis of ATP yields ADP and a phosphate group.
Function: Synthesis of carbohydrates and proteins, muscle contraction.
Importance: ATP is the energy currency of the cell.
