 Back
BackProteins: Structure, Function, Digestion, and Health Implications
Study Guide - Smart Notes

Proteins: Structure, Function, Digestion, and Health Implications
Introduction to Proteins
Proteins are essential macronutrients found in every cell of the human body. They play critical roles in cellular structure, function, and regulation. Unlike carbohydrates and lipids, proteins are composed of amino acids, which are linked together in unique sequences determined by DNA.
Definition: Proteins are large, complex molecules made up of amino acids and are necessary for the structure, function, and regulation of the body's tissues and organs.
Examples: Enzymes, hormones, antibodies, and structural components like muscle fibers.
Classification and Structure of Amino Acids
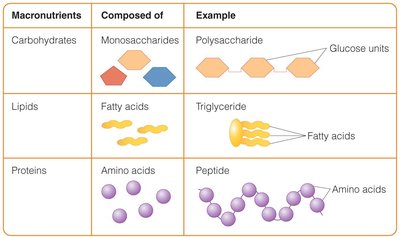
Amino acids are the building blocks of proteins. There are 20 different amino acids, which can be classified as essential, nonessential, or conditionally essential. The sequence and number of amino acids determine the protein's structure and function.
Essential amino acids: Must be obtained from the diet (9 types).
Nonessential amino acids: Can be synthesized by the body (11 types).
Conditionally essential amino acids: Normally nonessential but become essential under certain conditions (e.g., illness).
Peptides: Short chains of amino acids (dipeptides, tripeptides, polypeptides).
Proteins: Polypeptides with more than 50 amino acids.
Protein Structure and Denaturation
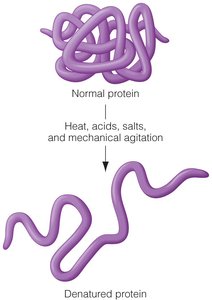
Proteins have four levels of structure: primary, secondary, tertiary, and quaternary. The shape of a protein is crucial for its function. Denaturation is the process by which proteins lose their structure due to external stress, such as heat or acid, which can alter their function but not their primary sequence.
Primary structure: Linear sequence of amino acids.
Secondary structure: Local folding into alpha-helices or beta-sheets.
Tertiary structure: Three-dimensional folding driven by interactions among side chains.
Quaternary structure: Association of multiple polypeptide chains.
Denaturation: Unfolding of proteins due to heat, acids, bases, salts, or mechanical agitation.
Digestion and Absorption of Proteins
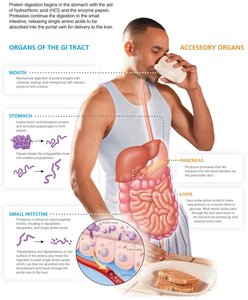
Protein digestion begins in the stomach and continues in the small intestine. Enzymes break down proteins into amino acids, which are then absorbed and transported to the liver for further metabolism or distribution to body tissues.
Stomach: Gastrin stimulates HCl release, denaturing proteins and activating pepsinogen to pepsin, which breaks polypeptides into smaller chains.
Small intestine: Pancreatic enzymes (trypsin, chymotrypsin, carboxypeptidase) and brush border enzymes (aminopeptidase, tripeptidase, dipeptidase) further digest peptides into amino acids.
Absorption: Amino acids are absorbed into the bloodstream and transported to the liver via the portal vein.
Metabolism of Amino Acids
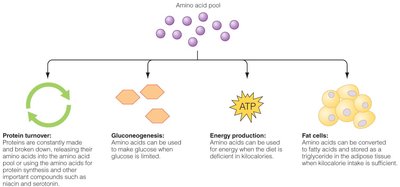
The liver plays a central role in amino acid metabolism. Amino acids can be used for protein synthesis, converted to glucose (gluconeogenesis), used for energy, or stored as fat if consumed in excess.
Protein turnover: Continuous breakdown and synthesis of proteins in the body.
Gluconeogenesis: Conversion of amino acids to glucose when carbohydrate intake is low.
Energy production: Amino acids can be used for ATP production when energy is needed.
Fat storage: Excess amino acids can be converted to fatty acids and stored as triglycerides.
Functions of Proteins in the Body
Proteins serve a wide range of functions, including structural support, catalysis, transport, immune defense, and regulation of fluid and acid-base balance. They also provide energy and contribute to satiety.
Structural support: Collagen in skin, keratin in hair and nails.
Enzymes: Catalyze biochemical reactions.
Hormones: Regulate physiological processes (e.g., insulin).
Fluid balance: Maintain proper distribution of fluids in body compartments.
Acid-base balance: Act as buffers to maintain pH.
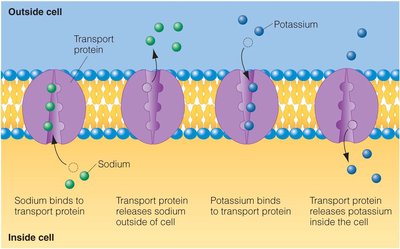
Transport: Carry substances like oxygen (hemoglobin) and ions across membranes.
Immune function: Antibodies defend against pathogens.
Energy: Provide 4 kcal/g when used as fuel.
Satiety: Promote fullness and appetite control.
Protein Requirements and Dietary Recommendations
Protein needs vary based on age, activity level, and physiological status. The Recommended Dietary Allowance (RDA) for adults is 0.8 g/kg of body weight per day, with an Acceptable Macronutrient Distribution Range (AMDR) of 10–35% of total daily calories from protein.
RDA: body weight per day for adults over 18.
AMDR: 10–35% of total daily kilocalories.
Athletes: May require 50–100% more protein due to increased demands.
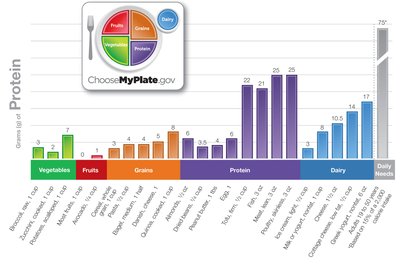
Food Sources and Quality of Protein
Proteins vary in quality based on their amino acid composition and digestibility. Animal sources are typically complete proteins, while most plant sources are incomplete but can be combined to provide all essential amino acids.
Complete proteins: Contain all nine essential amino acids (e.g., meat, eggs, dairy, quinoa, chia).
Incomplete proteins: Lacking one or more essential amino acids (e.g., most plant proteins).
Complementary proteins: Combining plant foods to provide all essential amino acids (e.g., beans and rice).
Best sources: Eggs, meat, fish, dairy, legumes, nuts, seeds.
Supplements: Generally unnecessary with a balanced diet.
Health Consequences of Protein Imbalance
Both excessive and inadequate protein intake can have significant health consequences. Excess protein may increase the risk of heart disease, kidney stones, and osteoporosis, while inadequate intake can lead to protein-energy malnutrition (PEM), impaired immunity, and poor growth.
Too much protein: Increased risk of heart disease (especially from animal sources high in saturated fat), kidney stones, and potential bone loss if calcium intake is low.
Too little protein: Muscle wasting, impaired immune function, poor wound healing, and protein-energy malnutrition (PEM).
Types of Protein-Energy Malnutrition (PEM)
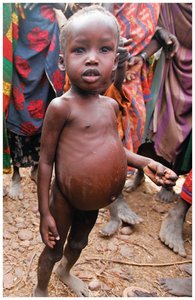
Kwashiorkor: Severe protein deficiency, often with adequate energy intake. Symptoms include edema, muscle wasting, and increased susceptibility to infection.
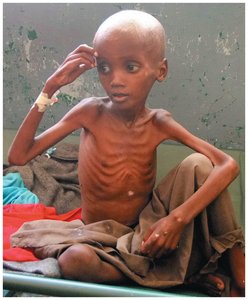
Marasmus: Severe deficiency of both protein and energy, leading to extreme wasting and frailty.
Treatment of PEM
Step 1: Address life-threatening factors (dehydration, nutrient imbalances).
Step 2: Gradually restore tissue with nutrient-dense foods and high-quality protein.
Step 3: Transition to regular foods and introduce physical activity.
Vegetarian Diets: Types, Benefits, and Risks
Vegetarian diets focus on plant-based foods and may exclude some or all animal products. While these diets can reduce the risk of chronic diseases, careful planning is needed to avoid nutrient deficiencies.
Lacto-vegetarian: Includes dairy, excludes meat, fish, poultry, eggs.
Lacto-ovo-vegetarian: Includes dairy and eggs, excludes meat, fish, poultry.
Ovo-vegetarian: Includes eggs, excludes dairy, meat, fish, poultry.
Vegan: Excludes all animal products.
Pescetarian: Includes fish, dairy, eggs; excludes meat, poultry.
Semivegetarian: Occasionally includes meat, fish, poultry.
Benefits: Lower risk of heart disease, hypertension, diabetes, cancer, stroke, and obesity.
Risks: Potential deficiencies in protein, calcium, iron, vitamin B12, zinc, vitamins A and D, and omega-3 fatty acids if not well planned.
