 Back
BackAtoms and the Periodic Table: Structure, Properties, and Quantum Models
Study Guide - Smart Notes

Atoms Are Ancient and Empty
Origin and Structure of Atoms
Atoms are the fundamental building blocks of matter, dating bgack to the birth of the universe. Most atoms are composed of empty space, with their mass concentrated in the nucleus. Elements heavier than hydrogen and much of the helium were produced in the interiors of stars.
Ancient Atoms: Most atoms have existed since the universe's formation.
Empty Space: Atoms are mostly empty, with electrons orbiting a dense nucleus.
Stellar Nucleosynthesis: Heavier elements are formed in stars.
The Elements
Definition and Classification
An element is a material made of only one kind of atom. The term "element" is used for macroscopic quantities, while "atom" refers to the submicroscopic unit.
Atom: The fundamental unit of an element.
Element: Any material consisting of only one type of atom.
Distinct Atoms: There are 115 known kinds of atoms, 90 found in nature, others synthesized.
Protons and Neutrons
Atomic Structure
Atoms consist of protons, neutrons, and electrons.
Protons: Positively charged, about 1800 times as massive as electrons.
Neutrons: Electrically neutral, similar mass to protons.
Electrons: Negatively charged, identical, repel each other, prevent atomic closeness.
Nucleons: Protons and neutrons together are called nucleons.
Atomic Number and Isotopes
Atomic Number: Number of protons in the nucleus; determines the element.
Isotopes: Atoms of the same element with different numbers of neutrons.
Mass Number: Total number of protons and neutrons in the nucleus.
Atomic Mass: Total mass of the atom, listed in atomic mass units (amu).
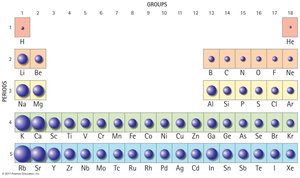
The Periodic Table
Organization and Structure
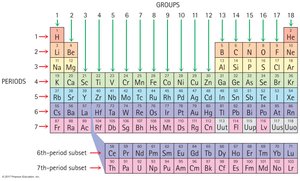
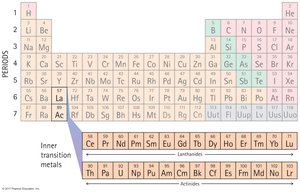
The Periodic Table is a systematic listing of all known elements, organized by increasing atomic number.
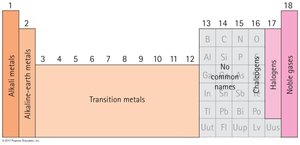
Groups: Vertical columns, elements with similar properties.
Periods: Horizontal rows, elements with increasing atomic number.
Reading the Table: Chemists use the table as a reference, not for memorization.
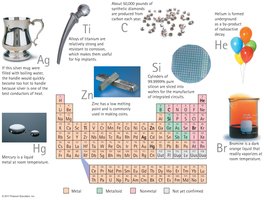
Element Classification
Elements are classified as metals, nonmetals, and metalloids.
Metals: Good conductors, malleable, ductile.
Nonmetals: Poor conductors, brittle.
Metalloids: Intermediate properties.
Atomic Size Trends
Atomic size varies across the periodic table.
Trend: Atoms generally decrease in size across a period and increase down a group.
Example: Lithium atom is larger than fluorine atom; arsenic atom is larger than sulfur atom.
Physical and Conceptual Models
Model Types
Physical Model: Replicates the object at a convenient scale.
Conceptual Model: Describes a system, such as the atom.
Atoms: Best described by conceptual models due to their submicroscopic nature.
Identifying Atoms Using the Spectroscope
Spectroscopy and Atomic Spectra
A spectroscope separates light into its component frequencies, allowing identification of elements by their unique spectral patterns.
Atomic Spectrum: Each element emits a distinctive pattern of discrete frequencies when energized.
Fingerprint: The atomic spectrum acts as a fingerprint for each element.
Historical Discoveries: Hydrogen's spectrum is more orderly; mathematical relationships found by Balmer and Rydberg.

Atomic Excitation
When atoms are excited, electrons are boosted to higher energy levels.
Energy Levels: Electrons can occupy multiple energy levels, leading to many spectral lines.
Standing Waves: Many standing electron waves can fit in the shell of the hydrogen atom.
The Quantum Hypothesis
Quantization of Energy
Max Planck proposed that radiant energy is emitted in discrete bundles called quanta. Einstein extended this to light, introducing the concept of the photon.
Energy of Photon: where is energy, is Planck's constant, and is frequency.
Wave-Particle Duality: Light exhibits both wave and particle properties.
Energy-Frequency Relationship: Higher frequency light has greater energy per photon.
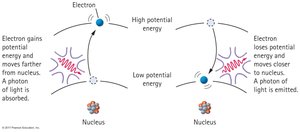
Bohr Model and Energy Levels
Niels Bohr explained atomic spectra using the quantum hypothesis.
Energy Levels: Electrons occupy discrete energy levels, each with a principal quantum number (integer).
Transitions: Electrons absorb energy to move to higher levels and emit photons when dropping to lower levels.
Ground State: The lowest energy level; electrons cannot move closer to the nucleus.

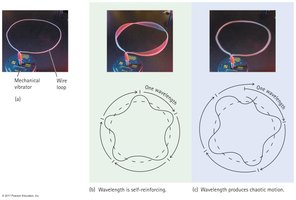
Electron Waves
Wave Nature of Electrons
Electrons in atoms behave as standing waves, restricting them to particular energy levels.
Standing Waves: Only integral numbers of wavelengths fit in the electron's orbit.
Energy Levels: is one wavelength, is two wavelengths, etc.
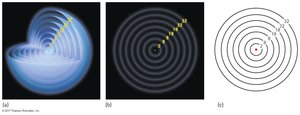
The Shell Model
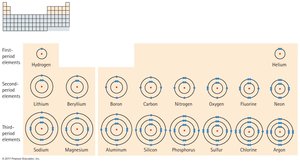
Electron Shells and Periods
The shell model describes electron arrangement in atoms, with shells corresponding to energy levels.
Shells: Electrons occupy shells around the nucleus.
Periods: Each period in the periodic table corresponds to a new shell.
Summary Table: Atomic Structure and Periodic Table
Concept | Definition | Example/Application |
|---|---|---|
Element | Material made of one kind of atom | Gold (Au), Hydrogen (H) |
Atom | Fundamental unit of an element | Single hydrogen atom |
Proton | Positively charged nucleon | Atomic number = number of protons |
Neutron | Neutral nucleon | Isotopes differ by neutron number |
Electron | Negatively charged particle | Electron transitions emit photons |
Isotope | Same element, different neutrons | Carbon-12, Carbon-14 |
Atomic Mass | Total mass of atom | Listed in amu |
Group | Vertical column in periodic table | Alkali metals, Halogens |
Period | Horizontal row in periodic table | Period 2: Li, Be, B, C, N, O, F, Ne |
Quantum Hypothesis | Energy quantized in photons | |
Shell Model | Electron arrangement in shells | First three periods |
Additional info: Academic context was added to clarify atomic structure, quantum models, and periodic table organization for physics students.
