 Back
BackChapter 18: A Macroscopic Description of Matter – Study Notes
Study Guide - Smart Notes

Chapter 18: A Macroscopic Description of Matter
Introduction to Macroscopic Systems
Macroscopic systems are collections of matter large enough to be observed and measured directly. Their properties, such as volume, density, pressure, and temperature, are often explained by the microscopic motions and interactions of atoms and molecules. Understanding these properties is essential for fields ranging from basic science to engineering.
Phases of Matter
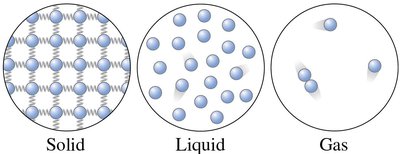
Solids, Liquids, and Gases
Most materials exist in three common phases: solid, liquid, and gas. Each phase has distinct macroscopic and microscopic characteristics.
Solid: Rigid structure; atoms vibrate around fixed equilibrium positions, connected by spring-like molecular bonds.
Liquid: Nearly incompressible; molecules are close together but free to move, allowing the liquid to flow and take the shape of its container.
Gas: Highly compressible; molecules move freely and interact only during occasional collisions.
Phase Changes
Phase changes occur when matter transitions between solid, liquid, and gas due to changes in temperature and pressure. During these changes, energy is absorbed or released, but temperature remains constant while the phase transition occurs.
Melting: Solid to liquid
Freezing: Liquid to solid
Boiling: Liquid to gas
Condensation: Gas to liquid



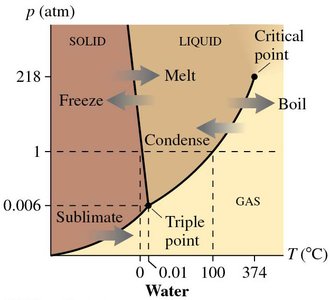
Phase Diagrams
Phase diagrams show the regions of stability for each phase as a function of temperature and pressure. The boundaries indicate conditions where two phases coexist in equilibrium. The triple point is where all three phases coexist.
Macroscopic Properties
Density
Density is the ratio of mass to volume for a material. It is a fundamental property used to characterize substances.
Formula:
SI Units: kg/m3
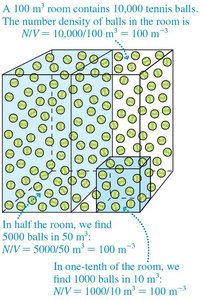
Number Density
Number density quantifies the number of atoms or molecules per unit volume.
Formula:
SI Units: m–3
Atomic Mass and Atomic Mass Number
The mass of an atom is primarily determined by its protons and neutrons. The atomic mass number (A) is the sum of protons and neutrons. The atomic mass unit (u) is defined as:
Atomic mass unit:
Atomic mass number:
Moles and Molar Mass
A mole is a standard unit for counting particles in chemistry and physics. One mole contains Avogadro’s number () of particles.
Avogadro’s number:
Number of moles:
Molar mass: (kg/mol) is the mass of 1 mole of substance.
Number of moles from mass:

Temperature and Thermal Properties
Temperature Scales
Temperature is a measure of the thermal energy in a system. Common scales include Celsius, Kelvin, and Fahrenheit.
Celsius to Kelvin:
Fahrenheit to Celsius:
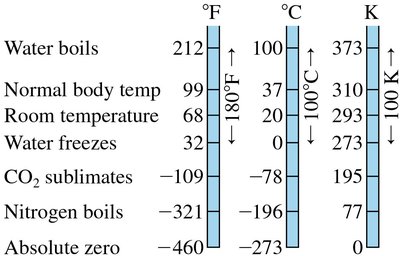
Absolute Zero
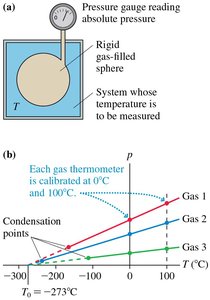
Absolute zero is the lowest possible temperature, where the pressure of an ideal gas extrapolates to zero. It forms the basis for the Kelvin scale.
Absolute zero:
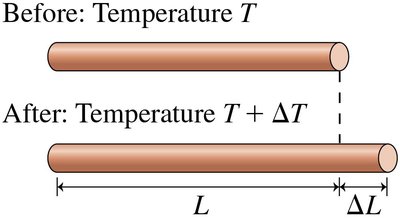
Thermal Expansion
Materials expand when heated. The change in length or volume is proportional to the temperature change.
Linear expansion:
Volume expansion:
For solids:
Ideal Gases and Gas Laws
Ideal-Gas Model
The ideal-gas model treats gas molecules as hard spheres that move freely and collide elastically. The model is valid at low densities and high temperatures.
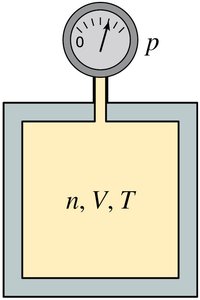
The Ideal-Gas Law
The state variables of an ideal gas—pressure (p), volume (V), number of moles (n), and temperature (T)—are related by the ideal-gas law:
Universal gas constant:
Boltzmann's constant:
Gas Processes and pV Diagrams
Gas processes can be visualized on pV diagrams, which plot pressure versus volume. Three basic processes are:
Isochoric (constant volume): Pressure changes with temperature.
Isobaric (constant pressure): Volume changes with temperature.
Isothermal (constant temperature): Pressure and volume change, but temperature remains fixed.
Summary Table: Densities of Various Materials
This table compares the densities of common substances.
Substance | Density (kg/m3) |
|---|---|
Air at STP | 1.29 |
Ethyl alcohol | 790 |
Water (solid) | 920 |
Water (liquid) | 1000 |
Aluminum | 2700 |
Copper | 8920 |
Gold | 19,300 |
Iron | 7870 |
Lead | 11,300 |
Mercury | 13,600 |
Silicon | 2330 |
Summary Table: Coefficients of Linear and Volume Expansion
This table lists the coefficients of linear and volume expansion for selected materials.
Material | Linear Expansion (α, °C–1) | Volume Expansion (β, °C–1) |
|---|---|---|
Aluminum | 2.3 × 10–5 | |
Brass | 1.9 × 10–5 | |
Concrete | 1.2 × 10–5 | |
Steel | 1.1 × 10–5 | |
Invar | 0.09 × 10–5 | |
Gasoline | 9.6 × 10–4 | |
Mercury | 1.8 × 10–4 | |
Ethyl alcohol | 1.1 × 10–4 |
Applications and Importance
Macroscopic properties are crucial for understanding and designing systems in physics, chemistry, biology, and engineering. Devices such as engines, power plants, and spacecraft rely on the behavior of materials under changing conditions of temperature and pressure.
Key Equations
Density:
Number density:
Atomic mass unit:
Number of moles:
Moles from mass:
Ideal-gas law:
Ideal-gas law (molecules):
Linear expansion:
Volume expansion:
Celsius to Kelvin:
Examples and Applications
Thermal expansion of pipes: Engineering must account for expansion and contraction due to temperature changes.
Gas pressure calculations: The ideal-gas law is used to determine pressure, volume, or temperature in sealed containers.
Phase changes: Cooking at high altitudes is affected by the lower boiling point of water.
Additional info: These notes expand on the original slides and examples, providing context and definitions for key terms and equations. All tables are recreated for clarity and completeness.
