 Back
BackChapter 19: Work, Heat, and the First Law of Thermodynamics – Study Notes
Study Guide - Smart Notes

Work, Heat, and the First Law of Thermodynamics
Introduction to the First Law of Thermodynamics
The first law of thermodynamics is a fundamental principle of energy conservation in physics. It states that the change in a system’s thermal energy is equal to the sum of energy transferred as work and as heat. This law is crucial for understanding energy exchanges in physical systems, especially in thermodynamic processes.
First Law Equation:
Energy Conservation: Energy cannot be created or destroyed, only transferred or transformed.
Applications: Used in engines, power plants, and spacecraft.
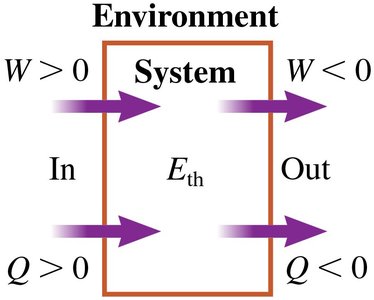
Energy Transfer: Work and Heat
Energy can be transferred to or from a system either as work or as heat. Work is associated with mechanical interactions, while heat is associated with thermal interactions due to temperature differences.
Work (W): Transfer of energy via mechanical means (forces causing displacement).
Heat (Q): Transfer of energy due to temperature difference between system and environment.
Sign Conventions:
W > 0: Energy added to system (compression)
W < 0: Energy extracted from system (expansion)
Q > 0: Energy added (environment hotter than system)
Q < 0: Energy extracted (system hotter than environment)
Mechanisms of Heat Transfer
Heat can be transferred between a system and its environment through several mechanisms:
Conduction: Direct transfer through material.
Convection: Transfer via fluid motion.
Radiation: Transfer via electromagnetic waves.
Evaporation: Transfer via phase change.
Thermal Properties of Matter
Materials respond to heat in ways governed by their thermal properties, such as specific heat, heat of fusion, heat of vaporization, and thermal conductivity. Calorimetry is the process of determining final temperature when systems interact thermally.
Specific Heat (c): Energy required to raise 1 kg of substance by 1 K.
Heat of Fusion (Lf): Energy required for phase change (solid to liquid).
Heat of Vaporization (Lv): Energy required for phase change (liquid to gas).
Thermal Conductivity (k): Measure of how well a material conducts heat.
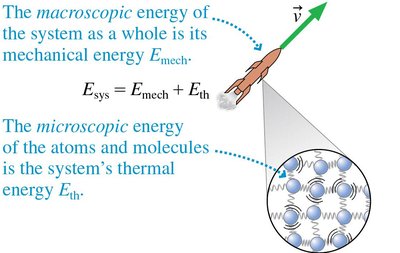
Energy Review and Conservation
The total energy of a system includes both macroscopic (mechanical) and microscopic (thermal) energy. For isolated systems, the total energy remains constant.
Energy Equation:
Isolated System: implies constant total energy.
Work in Ideal-Gas Processes
Work Done on a Gas
Work is done on a gas by changing its volume, typically using a piston. The mathematical expression for work in an ideal-gas process is:
Work Formula:
Geometric Interpretation: Work equals the negative area under the pV curve.

Sign of Work
The sign of work depends on whether the gas is compressed or expanded:
Compression: W > 0 (energy added)
Expansion: W < 0 (energy extracted)

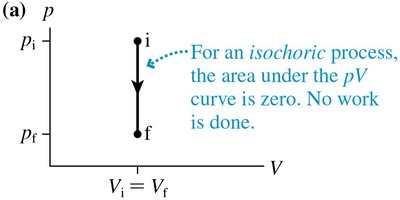
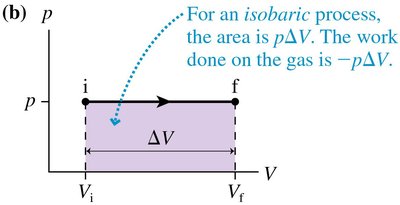
Special Ideal-Gas Processes
There are three important types of ideal-gas processes:
Isochoric Process: Volume constant, no work done ()
Isobaric Process: Pressure constant, work done is
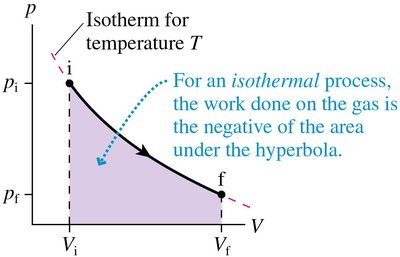
Isothermal Process: Temperature constant, work done is
Heat, Temperature, and Thermal Energy
Definitions and Relationships
Thermal energy is the energy due to the motion of atoms and molecules. Heat is energy transferred between system and environment, and temperature quantifies the system’s “hotness.”
Thermal Energy (Eth): Microscopic energy of moving molecules.
Heat (Q): Energy transferred due to temperature difference.
Temperature (T): State variable indicating thermal state.
Units of Heat
The SI unit of heat is the joule (J). Historically, the calorie was used:
1 cal = 4.186 J
1 food Calorie = 1 kcal = 4186 J
Understanding Work and Heat
Work and heat are two distinct ways of transferring energy. The following table summarizes their differences:
Work | Heat | |
|---|---|---|
Interaction | Mechanical | Thermal |
Requires | Force & displacement | Temperature difference |
Process | Macroscopic pushes/pulls | Microscopic collisions |
Positive value | W > 0: Compression | Q > 0: Environment hotter |
Negative value | W < 0: Expansion | Q < 0: System hotter |
Equilibrium | Mechanical equilibrium | Thermal equilibrium |
Specific Heat and Calorimetry
Specific Heat
Specific heat is the amount of energy required to raise the temperature of a unit mass (or mole) of a substance by one degree.
Formula: (mass-specific)
Molar Specific Heat:
Table: Specific Heats of Various Materials
Substance | c (J/kg K) | C (J/mol K) |
|---|---|---|
Aluminum | 900 | 24.3 |
Copper | 385 | 24.4 |
Iron | 449 | 25.1 |
Gold | 129 | 25.4 |
Lead | 128 | 26.5 |
Ice | 2090 | 37.6 |
Ethyl alcohol | 2400 | 110.4 |
Mercury | 140 | 28.1 |
Water | 4190 | 75.4 |
Calorimetry
Calorimetry is the study of heat transfer between interacting systems. When two or more systems interact thermally, they reach a common final temperature.
Energy Conservation:

Phase Changes and Heat of Transformation
Phase Change
During a phase change, thermal energy changes without a change in temperature. The heat required for a phase change is given by:
Formula:
Heat of Fusion (Lf): Melting/freezing
Heat of Vaporization (Lv): Boiling/condensing
Table: Melting/Boiling Temperatures and Heats of Transformation
Substance | Tm (°C) | Lf (J/kg) | Tb (°C) | Lv (J/kg) |
|---|---|---|---|---|
Nitrogen (N2) | -210 | 0.26 × 105 | -196 | 1.99 × 105 |
Ethyl alcohol | -114 | 1.09 × 105 | 78 | 8.79 × 105 |
Mercury | -39 | 0.11 × 105 | 357 | 2.96 × 105 |
Water | 0 | 3.33 × 105 | 100 | 22.6 × 105 |
Lead | 328 | 0.25 × 105 | 1750 | 8.58 × 105 |
Heat Transfer Mechanisms
Conduction
Conduction is the transfer of heat through a material. The rate of heat transfer is given by:
Formula:
Thermal Conductivity (k): Higher k means better conduction.
Table: Thermal Conductivities
Material | k (W/m K) |
|---|---|
Diamond | 2000 |
Silver | 430 |
Copper | 400 |
Aluminum | 240 |
Iron | 80 |
Stainless steel | 14 |
Ice | 1.7 |
Concrete | 0.8 |
Glass | 0.8 |
Styrofoam | 0.035 |
Air (20°C, 1 atm) | 0.023 |
Convection
Convection is the transfer of thermal energy by the motion of fluids. Heated fluid becomes less dense and rises, while cooler fluid sinks.
Radiation
Radiation is the transfer of energy via electromagnetic waves. The rate of heat transfer by radiation is:
Formula:
Emissivity (e): Ranges from 0 (no emission) to 1 (perfect emitter).
Stefan-Boltzmann Constant (σ): W/m2K4
Summary of Basic Gas Processes
Summary Table: Basic Gas Processes
Process | Work (W) | Heat (Q) | Thermal Energy Change (ΔEth) |
|---|---|---|---|
Isothermal | Nonzero | Nonzero | 0 |
Isochoric | 0 | Nonzero | Nonzero |
Adiabatic | Nonzero | 0 | Nonzero |
Important Concepts and Equations
First Law:
Work in Ideal-Gas Processes:
Calorimetry:
Adiabatic Process: ,
Heat of Transformation:
Specific Heat:
Molar Specific Heat:
Conduction:
Radiation:
Additional info: These notes expand on the preview and summary slides, providing definitions, formulas, and examples for each major concept in Chapter 19. All tables are recreated for clarity and completeness. Images are included only where they directly reinforce the explanation.
