 Back
BackChapter 31: Light Quanta – Quantum Theory and the Nature of Light
Study Guide - Smart Notes

Light Quanta and Quantum Theory
Birth of Quantum Theory
The nature of light has been debated for centuries, with arguments for both particle-like and wave-like behavior. The development of quantum theory resolved this debate by introducing the concept of quantization.
Wave Theory: Young's double-slit experiment (1801) demonstrated that light exhibits interference, a property of waves.
Quantum Hypothesis: In 1900, Max Planck proposed that radiant energy is emitted in discrete packets called quanta.
Photon: The quantum of light is called a photon.
Quantization and Planck's Constant
Quantum physics asserts that energy in atomic systems is quantized; only specific energy values are allowed. The energy of a photon is given by:
Planck's Equation:
Where E is energy, h is Planck's constant ( J·s), and f is the frequency of light.
Energy is always a whole-number multiple of this quantum value.
The Photoelectric Effect
Discovery and Explanation
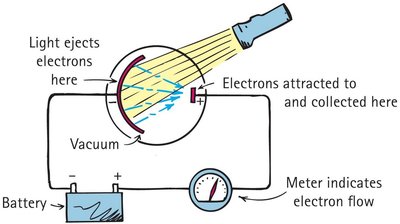
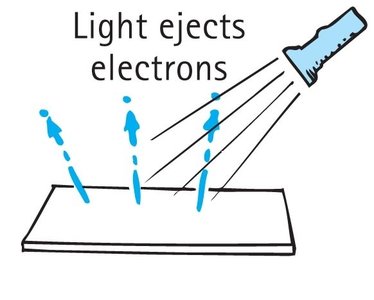
The photoelectric effect provided crucial evidence for the particle nature of light. When light shines on a photosensitive metal surface, electrons are ejected from the material.
Experimental Setup: Light strikes a metal surface, liberating electrons that are collected and measured as an electric current.
Key Observations:
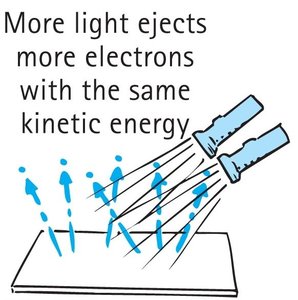
Brighter light (higher intensity) increases the number of ejected electrons, but not their velocity.
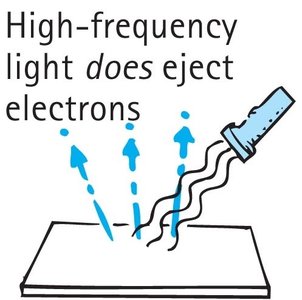
Higher frequency light increases the kinetic energy (velocity) of ejected electrons.
There is a threshold frequency below which no electrons are ejected, regardless of intensity.
Einstein's Interpretation: Light consists of photons, each with energy . A photon must have enough energy to dislodge an electron from the metal.

Key Questions and Answers
What happens if the light is brighter? More electrons are ejected, but their kinetic energy remains the same.
What happens if the light has a higher frequency? The ejected electrons have greater kinetic energy.
Wave–Particle Duality
Dual Nature of Light
Light exhibits both wave-like and particle-like properties, depending on the context of the experiment.
As a Particle: Light is absorbed or emitted as discrete photons (e.g., in the photoelectric effect).
As a Wave: Light exhibits interference and diffraction patterns (e.g., in the double-slit experiment).
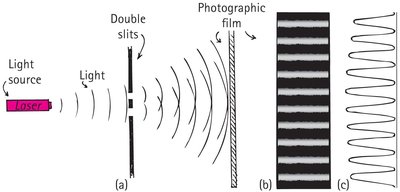
Double-Slit Experiment
Interference and the Nature of Photons
The double-slit experiment demonstrates the wave nature of light, even when photons pass through the slits one at a time.
Monochromatic light passing through two slits creates an interference pattern on a photographic film.
When the light is dimmed so that only one photon passes at a time, the interference pattern still emerges as more photons accumulate.
If one slit is covered, a single-slit diffraction pattern appears instead.
This experiment shows that each photon behaves as a wave while traveling and as a particle when detected.


Particles as Waves: Electron Diffraction
de Broglie Hypothesis
Louis de Broglie proposed that all matter exhibits wave-like properties. The wavelength associated with a particle is inversely proportional to its momentum:
Where is the wavelength, is Planck's constant, and is the momentum of the particle.
Electron diffraction experiments confirm that electrons (and other particles) can produce interference patterns, just like light.


Uncertainty Principle
Heisenberg's Uncertainty Principle
Werner Heisenberg formulated the uncertainty principle, which states that it is impossible to simultaneously know both the exact position and momentum of a particle. The more precisely one is known, the less precisely the other can be known:
Where is the uncertainty in momentum, is the uncertainty in position, and .
This principle also applies to energy and time: .
Uncertainty is significant only at the quantum (atomic) scale; it is negligible for macroscopic objects.
Complementarity
Bohr's Principle of Complementarity
Niels Bohr introduced the concept of complementarity to describe how quantum objects can display particle-like or wave-like properties depending on the experimental setup. This principle helps bridge the gap between the quantum world and everyday experience.
Modern physics uses the ideas of coherence and decoherence to further explain the transition between quantum and classical behavior.
