 Back
BackChapter 32: The Atom and the Quantum – Structured Study Notes
Study Guide - Smart Notes

The Atom and the Quantum
Discovery of the Atomic Nucleus
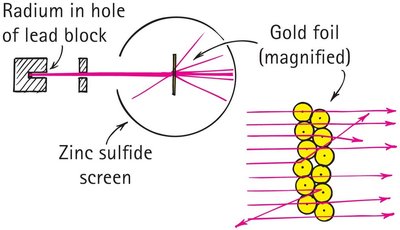
The atomic nucleus was discovered through Rutherford's gold foil experiment, which revealed the existence of a dense, positively charged core within the atom.
Alpha particles were directed at a thin gold foil; most passed through, but some were deflected or scattered backward.
Rutherford concluded that atoms contain a nucleus where most of the mass and positive charge are concentrated.
The experiment demonstrated that atoms are mostly empty space, with electrons orbiting the nucleus.
Example: The gold foil experiment is a classic demonstration of nuclear structure.
Discovery of the Electron
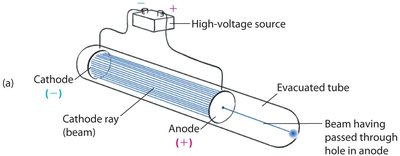
The electron was discovered using cathode ray tubes, which showed the existence of negatively charged particles.
Electrodes in a low-pressure tube produced a glowing ray from the cathode (negative terminal).
Slits and plates could manipulate the ray, demonstrating its particle nature.
Example: Cathode ray tubes are the basis for early television screens and oscilloscopes.
Deflection of Cathode Rays
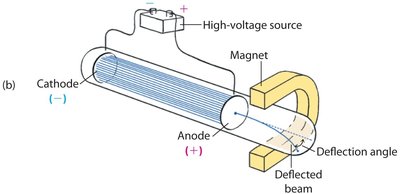
Further experiments showed that cathode rays could be deflected by electric and magnetic fields, confirming their negative charge.
The ray bent toward positive charges and away from negative charges.
Magnetic fields also caused deflection, indicating the ray consisted of charged particles.
Example: The deflection angle depends on the mass, charge, and speed of the particles.
Measurement of Electron Charge
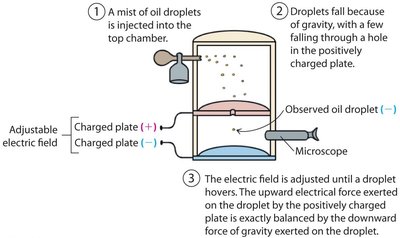
Millikan's oil drop experiment measured the charge of the electron by balancing gravitational and electrical forces on tiny oil droplets.
Oil droplets were sprayed into a chamber with charged plates.
The electric field was adjusted so droplets hovered, balancing gravity and electrical force.
Millikan found the charge on each drop was a multiple of a fundamental value—the electron's charge.
Formula: The force balance is (where is charge, is electric field, is mass, is gravity).
Atomic Spectra: Clues to Atomic Structure
Atomic spectra provide evidence for quantized energy levels in atoms, especially in hydrogen.
Hydrogen's spectrum shows a sequence of lines in the red, blue, violet, and ultraviolet regions.
Spacing between lines decreases as wavelength shortens, eventually merging in the ultraviolet.
Example: The Balmer series describes visible spectral lines of hydrogen.

Mathematical Formulas for Spectral Lines
Balmer and Rydberg developed formulas to describe the wavelengths and frequencies of spectral lines.
Balmer's formula successfully described hydrogen's visible lines.
Rydberg and Ritz extended these relationships to other elements, leading to the Ritz combination principle.
Formula: The Balmer formula for hydrogen is , where is an integer greater than 2.
Example: The sum of frequencies of two lines often equals the frequency of a third line.
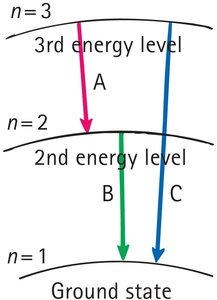
Bohr's Model of the Atom
Bohr proposed that electrons occupy discrete energy levels and emit light when transitioning between these levels.
Electrons reside in "stationary" states of fixed energy, not fixed position.
Quantum jumps between energy states result in emission or absorption of light.
The frequency of emitted radiation is determined by , where is energy difference, is Planck's constant, and is frequency.
Example: Light emission occurs when an electron drops from a higher to a lower energy level.
Classical Theory vs. Bohr Model
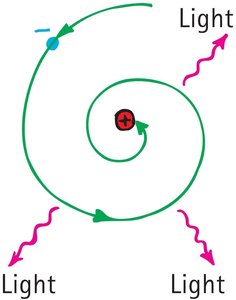
Classical physics predicted that electrons should spiral into the nucleus, but this does not occur due to quantized energy levels.
Accelerating electrons should emit radiation and lose energy, causing them to collapse into the nucleus.
Bohr's model prevents this by restricting electrons to specific orbits.
Example: Atoms remain stable because electrons cannot exist in arbitrary orbits.
Explanation of Quantized Energy Levels: Electron Waves
Louis de Broglie proposed that particles, including electrons, have associated wave properties, leading to quantized orbits.
The wavelength of a matter wave is inversely related to momentum: .
Bohr orbits exist where electron waves form standing waves, closing on themselves constructively.
Example: Electron waves in atoms are analogous to standing waves on a string.
Standing Wave Model of Electron Orbits
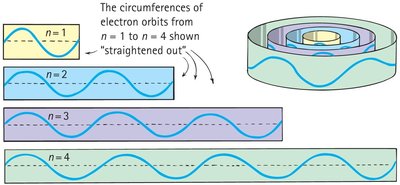
Electron orbits are allowed only where the circumference is a whole-number multiple of the electron's wavelength.
Constructive interference occurs when the wave closes in phase; destructive interference prevents other orbits.
Electron mass and charge are spread out as a standing wave around the nucleus.
Example: The innermost orbit has a circumference equal to one wavelength; the next has two, and so on.
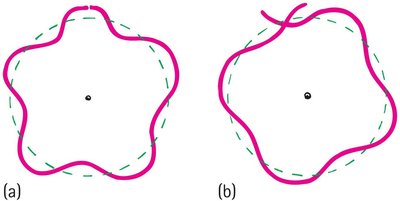
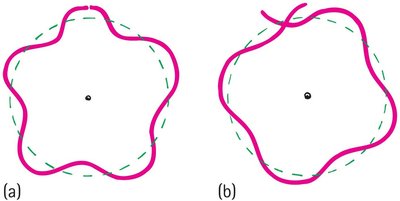
Discrete Energy States and Atomic Stability
Electron orbits have discrete radii and energy states because only whole-number multiples of wavelengths fit evenly into the orbit.
This explains why atoms do not shrink to the size of the nucleus.
No fraction of a wavelength is possible in a circular standing wave.
As long as electrons have the necessary momentum, atoms remain stable.
Quantum Mechanics
Quantum mechanics describes atomic and subatomic systems using wave functions and the Schrödinger equation.
The fundamental equation is the Schrödinger wave equation:
The wave function contains all information about the system.
Quantum mechanics generalizes the Bohr and de Broglie models, describing electrons as matter waves.
Example: The progression from Bohr's model to de Broglie waves to Schrödinger's model reflects increasing sophistication in atomic theory.
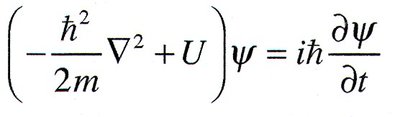
Correspondence Principle
The correspondence principle states that new theories must agree with old, verified theories in their common domain.
Formulated by Niels Bohr, it ensures continuity between classical and quantum physics.
Quantum mechanics reduces to classical mechanics for large systems or high quantum numbers.
Example: The Schrödinger equation leads to Newton's equations for orbital motion in the appropriate limit.
