 Back
BackChapter 38: Quantization – Study Notes for Physics for Scientists and Engineers
Study Guide - Smart Notes

Quantization: Introduction
Quantization is a fundamental concept in modern physics, describing how certain physical quantities, such as energy, can only take on discrete values rather than any value in a continuum. This chapter explores the quantization of energy for both light and matter, introducing the photon model of light, the wave-like nature of matter, and the quantum structure of atoms.
What is Quantization?
In classical physics, energy and other quantities are continuous. However, at the atomic scale, energy is quantized:
Photons: Light energy is divided into discrete packets called photons.
Quantum States: Electrons in atoms can only occupy certain energy levels, known as quantum states.
The Photon Model of Light
Definition and Properties
The photon model describes light as consisting of particle-like units called photons:
Each photon has energy , where is Planck's constant and is the frequency of light.
At low intensities, light is detected as discrete events, supporting the particle nature of light.

Consequences of the Photon Model
Photoelectric Effect: Short-wavelength light can eject electrons from a metal, but long-wavelength light cannot.
Compton Scattering: The wavelength of x-rays increases when scattered from a target, explained by photon collisions.
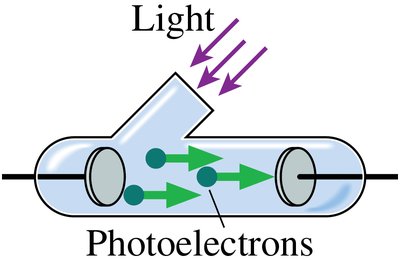
Photon Rate and Power
The total energy of photons:
Power delivered by light: , where is the photon arrival rate.
The Photoelectric Effect
Experimental Setup and Observations
When ultraviolet light shines on a metal cathode, electrons are emitted, creating a current. This is the photoelectric effect.
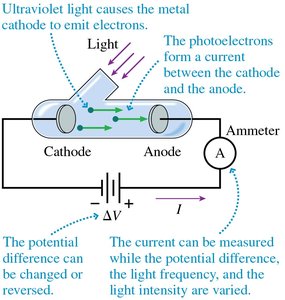
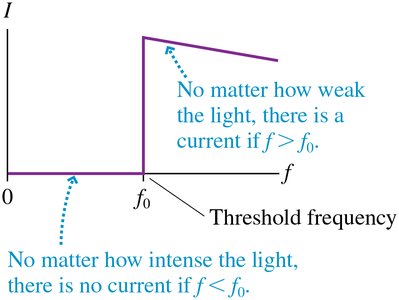
Photoelectrons are emitted only if the light frequency exceeds a threshold frequency .
The threshold frequency depends on the metal's work function .
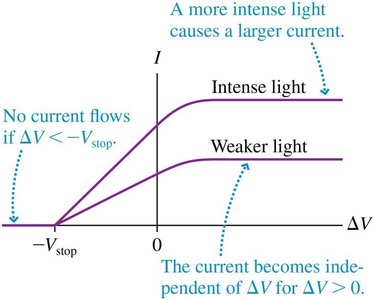
Stopping Potential and Work Function
Current decreases as the voltage between cathode and anode becomes negative, reaching zero at the stopping potential .
The work function is the minimum energy needed to free an electron from the metal.
Element | Work Function (eV) |
|---|---|
Potassium | 2.30 |
Sodium | 2.36 |
Aluminum | 4.28 |
Tungsten | 4.55 |
Copper | 4.65 |
Iron | 4.70 |
Gold | 5.10 |
Einstein’s Explanation
Light consists of photons, each with energy .
Electrons are ejected only if .
The maximum kinetic energy of ejected electrons: .
The stopping potential: .

Matter Waves and de Broglie Hypothesis
Wave-Particle Duality
Louis de Broglie proposed that particles such as electrons have wave-like properties, with a wavelength given by:
de Broglie wavelength:

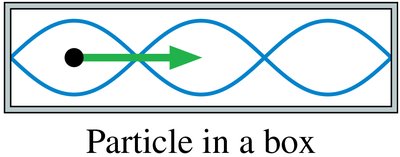
Particle in a Box
A particle confined in a one-dimensional box of length forms standing waves, leading to quantized energy levels:
Allowed wavelengths: ,
Allowed energies:
The Bohr Model of the Atom
Postulates and Energy Levels
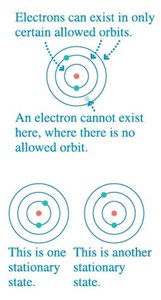
Electrons can only occupy certain allowed orbits (stationary states).
Each stationary state has a discrete energy .
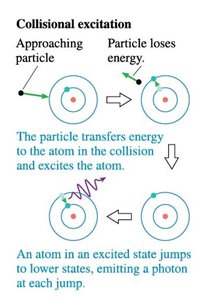
Transitions between states involve emission or absorption of photons with energy .


Quantization of Angular Momentum
Electron angular momentum is quantized:
Energy Levels in Hydrogen
Allowed radii: , where nm is the Bohr radius.
Allowed energies:
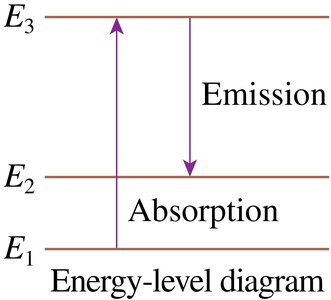
Energy-Level Diagrams and Spectra
Transitions between energy levels produce emission or absorption lines at specific wavelengths.
Applications and Important Concepts
Photoelectric Effect: Used to determine Planck's constant and the work function of materials.
Particle in a Box: Fundamental model for understanding quantum confinement and energy quantization.
Bohr Model: Explains the discrete spectra of hydrogen and other atoms.
Compton Scattering: Demonstrates the particle nature of light and conservation of energy and momentum in photon-electron collisions.
Summary Table: Key Equations
Concept | Equation |
|---|---|
Photon energy | |
de Broglie wavelength | |
Photoelectric effect (max KE) | |
Stopping potential | |
Particle in a box energy | |
Bohr energy levels (hydrogen) |
Conclusion
Quantization is a cornerstone of quantum physics, explaining phenomena that cannot be understood with classical physics. The photon model, matter waves, and quantized atomic energy levels form the basis for understanding modern physics and its applications in technology and nature.
