 Back
BackCoulomb’s Law and Electric Charge: Foundations of Electrostatics
Study Guide - Smart Notes

Coulomb’s Law and Electric Charge
Introduction to Electric Charge and Electrostatics
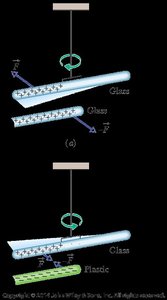
Electrostatics is the study of electric charges at rest and the forces they exert on each other. The fundamental property underlying these phenomena is the electric charge, which comes in two types: positive and negative. The interactions between charges are governed by the principles of attraction and repulsion: like charges repel, and opposite charges attract.
Electric charge is a physical property of matter responsible for electric phenomena.
Everyday devices such as computers and radios rely on the principles of electromagnetism, which unifies electric and magnetic effects.
Classification of Materials by Electrical Properties
Conductors, Insulators, Semiconductors, and Superconductors
Materials are classified based on their ability to allow the movement of electric charge:
Conductors: Materials (e.g., metals) with many free electrons that can move easily, allowing charge to flow.
Insulators (Nonconductors): Materials (e.g., rubber, glass) in which electrons are tightly bound and do not move freely.
Semiconductors: Materials (e.g., silicon, germanium) with electrical properties intermediate between conductors and insulators.
Superconductors: Materials that, below a certain temperature, allow charge to move without resistance.
Atomic Structure and Charge
Subatomic Particles and Their Charges
The electrical properties of materials are rooted in atomic structure. Atoms consist of a nucleus (protons and neutrons) surrounded by electrons. The movement of electrons, especially in conductors, is responsible for electrical conduction.
Particle | Symbol | Charge |
|---|---|---|
Electron | e or e- | -e |
Proton | p | +e |
Neutron | n | 0 |

Conduction electrons are the mobile electrons in a conductor that enable the flow of electric charge.
Electrostatic Forces and Coulomb’s Law
Basic Principles of Electrostatic Interaction
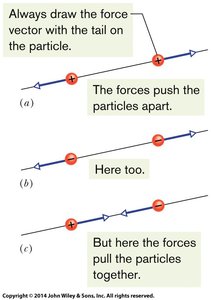
Electrostatic forces arise from the presence of electric charges. The direction of the force depends on the sign of the interacting charges:
Like charges (both positive or both negative) repel each other.
Opposite charges (one positive, one negative) attract each other.
Coulomb’s Law: Mathematical Formulation
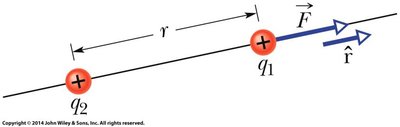
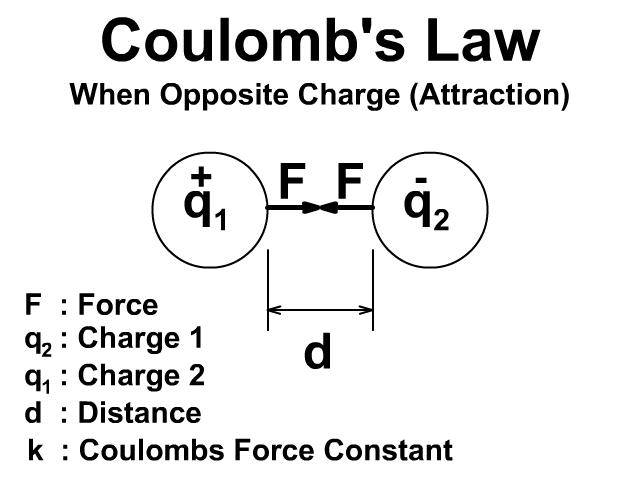
Coulomb’s Law quantifies the electrostatic force between two point charges. If two charges, and , are separated by a distance , the magnitude of the force between them is:
where is the permittivity of free space, and is the Coulomb constant.
Vector Nature of Electrostatic Force
The electrostatic force is a vector quantity. When multiple charges are present, the net force on any charge is the vector sum of the individual forces exerted by all other charges.
Forces are added as vectors, not scalars.
The direction of the force depends on the relative positions and signs of the charges.
Worked Example: Force Between Two Charges
Given two charges and separated by , the force is:
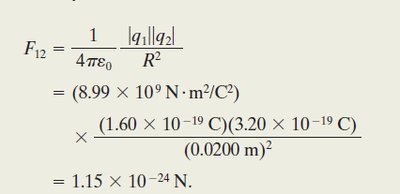
Worked Example: Net Force with Three Charges
When a third charge is introduced, calculate the force from each charge and sum the vectors:
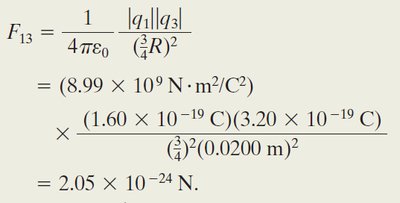
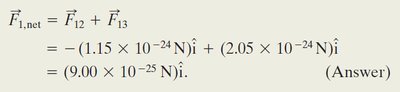
Induced Charge and Polarization
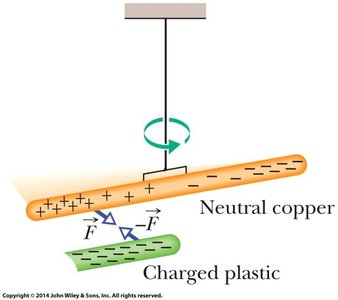
Induction and Redistribution of Charge
When a charged object is brought near a neutral conductor, it can induce a separation of charges within the conductor, leading to attraction even though the conductor is overall neutral. This is called induced charge.
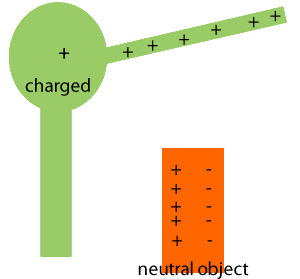
Quantization and Conservation of Charge
Quantization of Electric Charge
Electric charge is quantized, meaning it exists in discrete amounts. The smallest unit of charge is the elementary charge :
Any observable charge can be written as , where is an integer and .
Conservation of Electric Charge
The total electric charge in an isolated system is always conserved. Charge can be transferred from one object to another but cannot be created or destroyed.
When two objects are rubbed together, charge is transferred, not created.
In particle interactions, the sum of charges before and after the process remains the same.
Example: Electrostatic Force in a Nucleus
Calculate the repulsive force between two protons separated by in a nucleus:
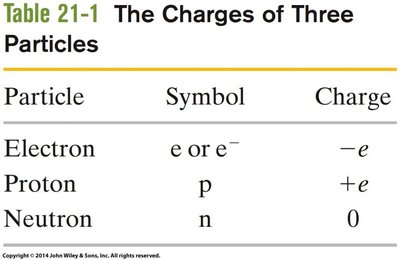
Summary Table: Charges of Subatomic Particles
Particle | Symbol | Charge |
|---|---|---|
Electron | e or e- | -e |
Proton | p | +e |
Neutron | n | 0 |

Key Points
Like charges repel; opposite charges attract.
Coulomb’s Law gives the force between two point charges.
Conductors allow free movement of charge; insulators do not.
Electric charge is quantized and conserved.
