 Back
BackEarly Quantum Theory and Models of the Atom
Study Guide - Smart Notes

Early Quantum Theory and Models of the Atom
J. J. Thomson’s e/m Experiment
J. J. Thomson’s experiment (c. 1897) was pivotal in determining the charge-to-mass ratio (e/m) of the electron. By passing an electron beam through perpendicular electric and magnetic fields, Thomson observed the deflection of the beam and derived the following relationship:
Key Equation: , where E is the electric field, B is the magnetic field, and r is the radius of curvature of the electron's path.
Result:
Observation: With both fields present and properly balanced, the electron beam is undeflected; with only the magnetic field, the beam curves.
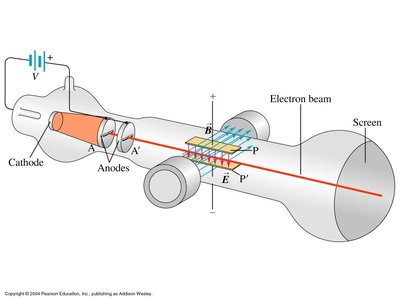
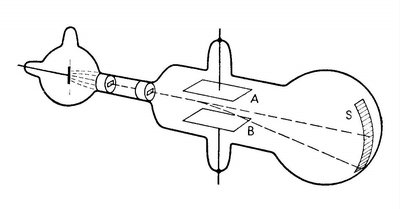
Example: This experiment established the existence of the electron as a particle with a specific charge-to-mass ratio, distinct from atoms.
Millikan Oil Drop Experiment
R. A. Millikan’s oil drop experiment (c. 1910) measured the elementary electric charge e by balancing the gravitational and electric forces on tiny charged oil droplets.
Key Equation:
Result: (smallest observed charge)
Mass of Electron: Using from Thomson and from Millikan,
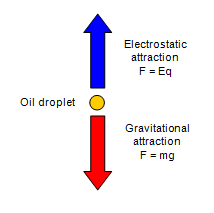
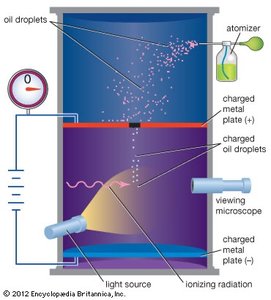
Example: The experiment demonstrated that electric charge is quantized and provided the first accurate value for the electron’s mass.
Blackbody Radiation and Planck’s Hypothesis
Thermal Radiation and Blackbody Concept
All objects emit electromagnetic radiation due to thermal motion of charged particles. A blackbody is an idealized object that absorbs all incident radiation and emits a characteristic spectrum dependent only on temperature.
Key Properties: The spectrum’s peak shifts to shorter wavelengths as temperature increases (Wien’s Law).
Wien’s Displacement Law:
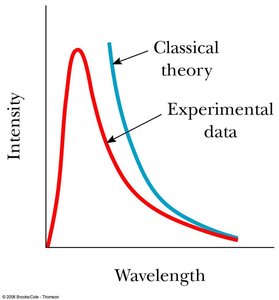
Ultraviolet Catastrophe: Classical physics predicted infinite energy at short wavelengths, which was not observed experimentally.
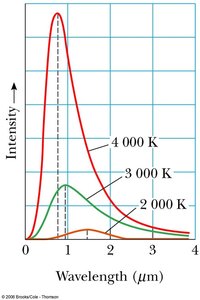
Example: The Sun’s surface temperature can be estimated from its peak emission wavelength using Wien’s Law.
Planck’s Quantum Hypothesis
To resolve the ultraviolet catastrophe, Max Planck proposed that energy is quantized and emitted in discrete packets called quanta.
Planck’s Formula: , where is a positive integer, is Planck’s constant (), and is frequency.
Implication: Only certain energy states are allowed, and higher frequency modes require more energy to excite.
Example: Calculating the temperature of stars or light bulb filaments using Wien’s Law and Planck’s quantization.
Photoelectric Effect and Particle Theory of Light
Photoelectric Effect
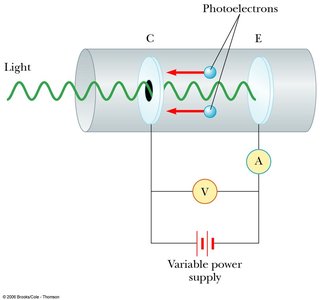
The photoelectric effect is the emission of electrons from a metal surface when illuminated by light of sufficient frequency. The emitted electrons are called photoelectrons.
Key Observations:
No electrons are emitted below a cutoff frequency, regardless of intensity.
The maximum kinetic energy of photoelectrons depends on light frequency, not intensity.
Emission is nearly instantaneous.
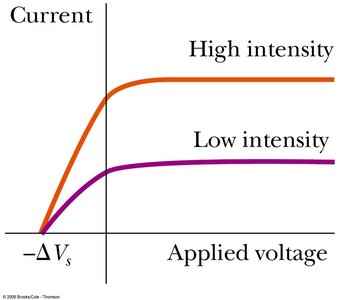
Example: The stopping potential is used to measure the maximum kinetic energy of photoelectrons: .
Einstein’s Particle Theory of Light
Einstein explained the photoelectric effect by proposing that light consists of particles called photons, each with energy .
Key Equation: , where is the work function of the metal.
Cutoff Frequency:
Predictions:
Photoelectrons are emitted only if .
increases linearly with frequency.
Emission is instantaneous if photon energy exceeds .
Example: For silver ( eV), the minimum frequency for photoemission is Hz.
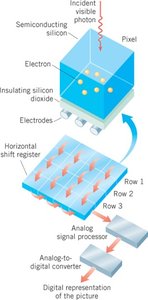
Applications: Charge-Coupled Devices (CCDs)
CCDs, used in digital cameras, operate based on the photoelectric effect. Incident photons liberate electrons, which are then processed to form digital images.
Photon Mass and Momentum
Photons are massless particles, but they carry energy and momentum. Special relativity provides the relationships:
Energy:
Momentum:
Example: A blue photon ( nm) has kg·m/s and eV.
Solar Sails: Application of Photon Momentum
Solar sails use the momentum of photons from sunlight to propel spacecraft. The force and resulting acceleration can be calculated using the photon flux and the sail’s mass per area.
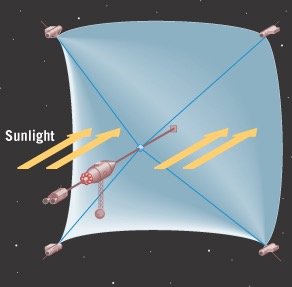
Example: A sail with mass per area kg/m2 and sunlight intensity W/m2 can reach speeds of after one year of acceleration.
Wave–Particle Duality
Light and matter exhibit both wave-like and particle-like properties. This duality is fundamental to quantum mechanics.
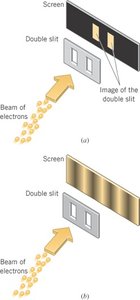
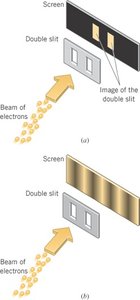
Wave Behavior: Interference and diffraction (e.g., double-slit experiment).
Particle Behavior: Photoelectric effect, photon momentum.
de Broglie Wavelength
Louis de Broglie proposed that all particles have a wavelength given by:
de Broglie Wavelength:
Example: An electron moving at m/s has m, comparable to atomic spacing, making interference observable. For a baseball, is negligibly small.
Neutron and X-ray Diffraction
Neutron and X-ray diffraction patterns provide evidence for the wave nature of particles, as observed in crystal structures.

Example: Neutrons with kinetic energy eV have a wavelength matching atomic spacing, allowing diffraction effects to be observed.
Probability Waves and Quantum Interference
Particles such as electrons are described by probability waves. In the double-slit experiment, even single electrons build up an interference pattern over time, demonstrating self-interference.

Additional info: This is a cornerstone of quantum mechanics, showing that the probability distribution, not deterministic paths, governs particle behavior at the quantum scale.
