 Back
BackElectric Charge and Coulomb’s Law: Fundamental Concepts and Experiments
Study Guide - Smart Notes

Electric Charge and Coulomb’s Law
Historical Development of Electricity
The study of electricity began with simple experiments involving materials such as amber and wool. Early scientists observed that rubbing certain materials could cause them to attract or repel other objects, leading to the identification of two types of electric charge. The concept evolved from the idea of two distinct fluids to a single fluid theory, with Benjamin Franklin popularizing the assignment of positive and negative charges.
Key Point 1: Early experiments showed that rubbed materials could attract or repel other objects.
Key Point 2: The distinction between positive and negative charges was established, with Franklin's convention still used today.
Example: Rubbing amber with wool causes it to attract bits of straw or feathers.

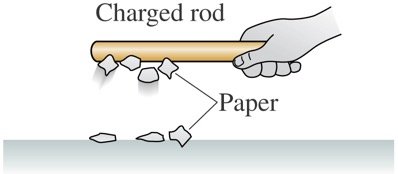
Charging Experiments: Forces Between Charges
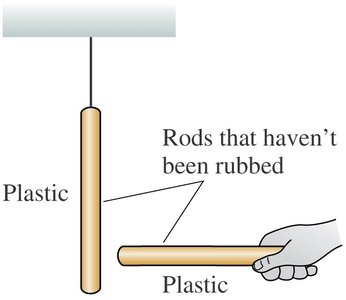
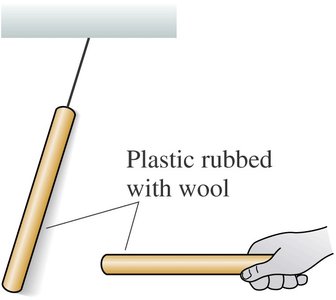
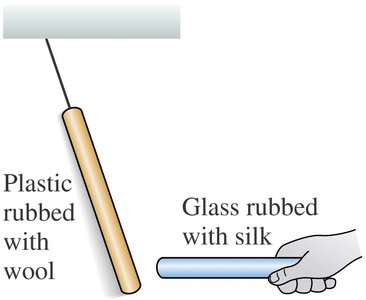
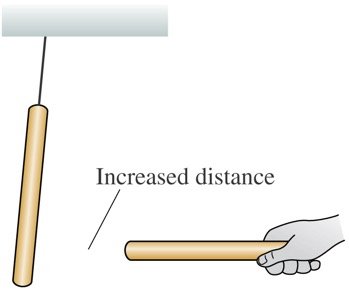
Experiments with plastic and glass rods demonstrate the fundamental properties of electric charge. When rods are rubbed with materials like wool or silk, they acquire charge and exhibit attractive or repulsive forces depending on the type of charge.
Key Point 1: Un-rubbed rods show no force between them.
Key Point 2: Like charges repel, unlike charges attract.
Key Point 3: The force decreases with increased distance between the charges.
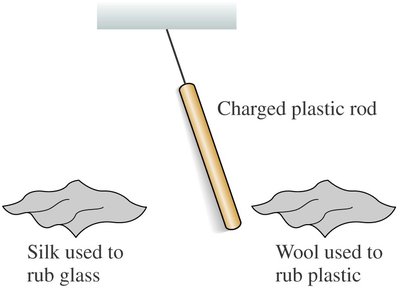
Example: A plastic rod rubbed with wool repels another similarly charged rod, but attracts a glass rod rubbed with silk.

Charging Experiments: Two Types of Charges
Further experiments reveal that charged objects interact differently with neutral and other charged objects. The process of charging by friction and the resulting behavior of objects help distinguish between positive and negative charges.
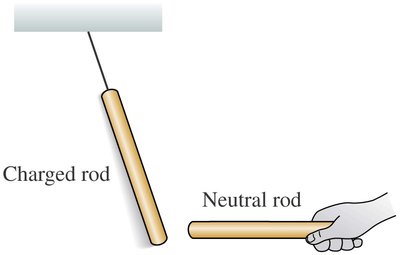
Key Point 1: Charged rods attract neutral objects.
Key Point 2: Charged rods are attracted to the material used for rubbing and repelled by the opposite material.
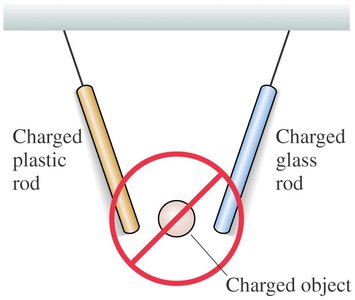
Key Point 3: No object attracts both a charged plastic rod and a charged glass rod simultaneously.
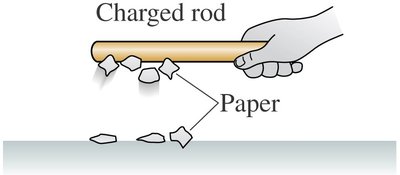
Example: A charged plastic rod attracts small pieces of paper and is attracted to an un-rubbed rod.

Charging Experiments: Transfer of Charges
Charge can be transferred from one object to another, especially when conductors are involved. The ability to transfer charge depends on the material connecting the objects.
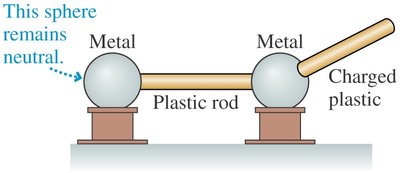
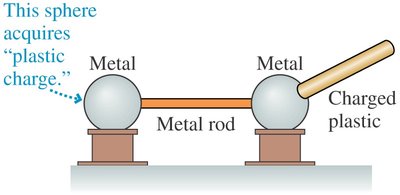
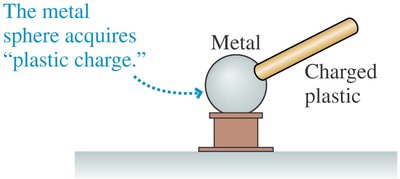
Key Point 1: Charge from a plastic rod can be transferred to a metal sphere.
Key Point 2: A second metal sphere connected by a metal rod acquires the charge, but not if connected by a plastic rod.
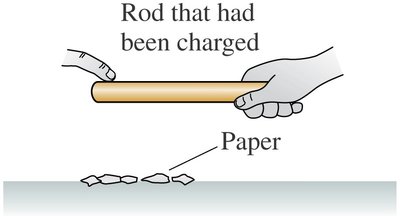
Example: After transferring charge, the rod no longer attracts paper, indicating loss of charge.

Charge Model
Charge Model, Part I
The charge model explains the behavior of electric charges and their interactions. Frictional forces can add or remove charge from objects, and there are only two types of charge: positive and negative.
Key Point 1: Frictional forces cause charging by separating charges.
Key Point 2: Like charges repel, opposite charges attract.
Key Point 3: Neutral objects have equal mixtures of both types of charge.
Example: Rubbing a rod separates the mixture of charges, creating a net charge.


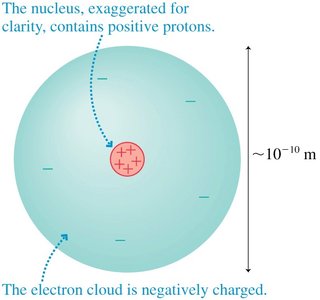
Atoms and Electricity
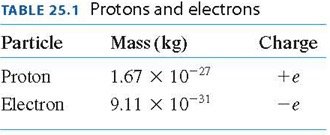
Atoms are composed of a positively charged nucleus and negatively charged electrons. The fundamental unit of charge is denoted by e, and protons and electrons have equal but opposite charges.
Key Point 1: The atom is held together by electric forces between the nucleus and electrons.
Key Point 2: The charge of a proton is +e, and the charge of an electron is -e.
Example: The atomic mass number is given by , where Z is the number of protons and N is the number of neutrons.

Particle | Mass (kg) | Charge |
|---|---|---|
Proton | 1.67 × 10-27 | +e |
Electron | 9.11 × 10-31 | -e |
Positive and Negative Ions
Atoms can become ions by gaining or losing electrons. A loss of electrons results in a positive ion, while a gain results in a negative ion.
Key Point 1: If the number of protons does not equal the number of electrons, the atom has a net charge.
Key Point 2: Positive ions have a net charge of , negative ions have .
Example: A neutral atom losing one electron becomes a positive ion.

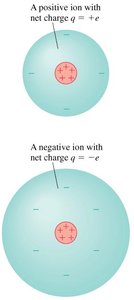
Charging Objects and Conservation of Charge
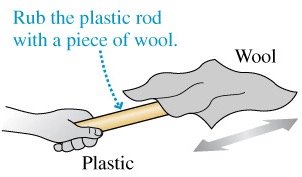
Charging by Friction
Friction can break molecular bonds and separate charges, creating positive and negative ions. This process is fundamental to understanding how objects become charged.
Key Point 1: Rubbing materials together causes charge separation.
Key Point 2: The process creates molecular ions with opposite charges.
Example: Rubbing a plastic rod with wool produces charge separation.

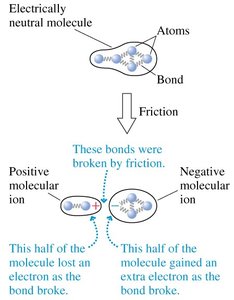
Conservation of Charge
Electric charge is conserved in all processes. The net charge of an isolated system remains constant, even as charges are separated or transferred.
Key Point 1: Charge cannot be created or destroyed.
Key Point 2: The sum of charges before and after any process is equal.
Example: Rubbing a plastic rod with wool results in equal and opposite charges on each.

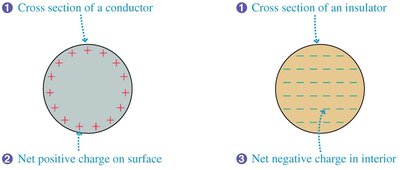
Conductors and Insulators
Conductors and Insulators: Properties
Materials are classified as conductors or insulators based on their ability to allow charge movement. Conductors allow charges to move freely, while insulators do not.
Key Point 1: In conductors, charge resides on the surface.
Key Point 2: In insulators, charge may reside in the interior and is immobile.
Example: Metals are conductors, plastics are insulators.

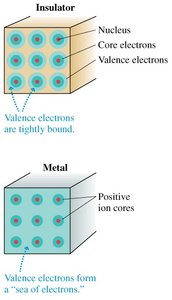
Microscopic View: Electrons in Materials
In insulators, electrons are tightly bound and immobile. In metals, valence electrons are loosely bound and form a "sea" of electrons, allowing for conduction.
Key Point 1: Electrons are the primary charge carriers in metals.
Key Point 2: Other materials, such as semiconductors and ionic liquids, have different charge carriers.
Example: Valence electrons in metals move freely, enabling electrical conduction.

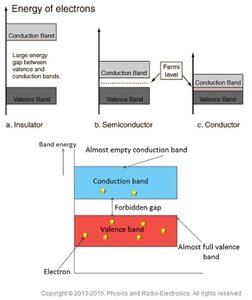
Conduction and Valence Bands
The energy bands in materials determine their conductive properties. Insulators have a large gap between valence and conduction bands, while conductors have overlapping bands.
Key Point 1: The band gap prevents electron movement in insulators.
Key Point 2: Conductors have electrons in the conduction band, allowing free movement.
Example: Semiconductors have a small band gap, allowing limited conduction.

Charging Insulators and Conductors
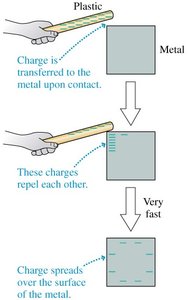
Charging Methods
Conductors cannot be charged by friction but can acquire charge by contact. Insulators can be charged by friction, with the charge remaining immobile.
Key Point 1: Charge on conductors spreads over the surface.
Key Point 2: Charge on insulators remains localized.
Example: Touching a metal object with a charged rod transfers charge to the metal.

Electroscope and Charge Detection
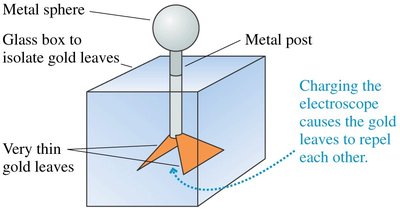
The Electroscope
An electroscope is an instrument used to detect electric charge. It operates by observing the movement of gold leaves due to electrostatic forces.
Key Point 1: Charging the electroscope causes the leaves to repel each other.
Key Point 2: The angle of deflection indicates the amount of charge.
Example: Touching a charged object to the electroscope transfers charge, causing leaf movement.

Charge Polarization and Induction
Charge Polarization
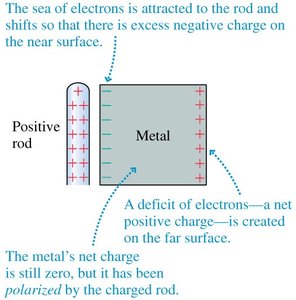
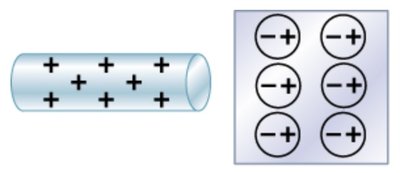
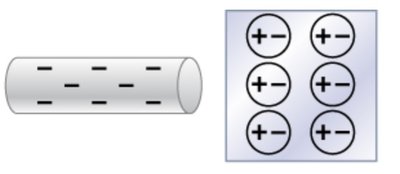
Charge polarization occurs when a charged object induces a redistribution of charges in a nearby conductor or insulator. This effect is used in charging by induction.
Key Point 1: Bringing a charged rod near a conductor causes electrons to shift, creating polarized surfaces.
Key Point 2: The net charge remains zero, but surfaces become oppositely charged.
Example: Induced charge can be observed by bringing a charged rod near an electroscope.

Charging by Induction
Charging by induction involves creating a net charge on an object without direct contact. This is achieved by polarizing the object and then separating it from the source of polarization.
Key Point 1: Induction relies on charge polarization and separation.
Key Point 2: The process results in one object acquiring positive charge and the other negative charge.
Example: Metal spheres can be charged by induction when a charged rod is brought near and the spheres are separated.
Additional info: The notes cover the foundational concepts of electric charge, materials, and Coulomb's Law, suitable for college-level physics students.
