 Back
BackElectric Charge and Electric Field – Study Notes
Study Guide - Smart Notes

Chapter 16: Electric Charge and Electric Field
16.1 Static Electricity; Electric Charge and Its Conservation
Static electricity arises when objects are charged by rubbing, resulting in the transfer of electric charge. Electric charge exists in two types: positive and negative. Like charges repel each other, while opposite charges attract. Importantly, electric charge is conserved, meaning the total charge in an isolated system remains constant during any interaction.
Static electricity: The accumulation of charge on the surface of objects.
Conservation of charge: The arithmetic sum of the total charge cannot change in any interaction.
16.2 Electric Charge in the Atom
An atom consists of a small, massive, positively charged nucleus surrounded by a cloud of negatively charged electrons. Atoms are electrically neutral because the number of protons equals the number of electrons. Rubbing objects can transfer electrons, causing them to become charged. Some molecules, called polar molecules, are neutral overall but have uneven charge distribution.
Nucleus: Contains protons (positive) and neutrons (neutral).
Electron cloud: Contains electrons (negative).
Polar molecule: Example: Water (H2O).
16.3 Insulators and Conductors
Materials are classified based on their ability to conduct electric charge:
Conductors: Charge flows freely (e.g., metals).
Insulators: Almost no charge flows (e.g., glass, rubber).
Semiconductors: Materials with conductivity between conductors and insulators.
16.4 Induced Charge; the Electroscope
Objects can be charged by conduction (direct contact) or induction (without contact). Nonconductors do not become charged by these methods but can experience charge separation. An electroscope is a device used to detect and determine the sign of electric charge.
Charging by conduction: Transfer of charge by direct contact.
Charging by induction: Redistribution of charge without direct contact.
Electroscope: Detects and identifies electric charge.
16.5 Coulomb’s Law
Coulomb’s law quantifies the electric force between two point charges. The force is proportional to the product of the charges and inversely proportional to the square of the distance between them. The force is attractive for opposite charges and repulsive for like charges.
Equation (magnitude):
Unit of charge: Coulomb (C)
Proportionality constant:
Charge quantization: (elementary charge)
Permittivity of free space: ,
Superposition principle: For multiple charges, add forces vectorially.
16.6 Solving Problems Involving Coulomb’s Law and Vectors
To find the net force on a charge, calculate the vector sum of all individual forces acting on it. Use vector addition techniques to combine forces from multiple charges.
Draw diagrams showing all charges and forces.
Apply Coulomb’s law for each pair of charges.
Add forces vectorially to find the net force.
16.7 The Electric Field
The electric field at a point is defined as the force per unit charge experienced by a small positive test charge placed at that point. The electric field produced by a point charge decreases with the square of the distance from the charge.
Definition:
Field of a point charge:
Force on a charge in an electric field:
Superposition principle: Add electric fields from all charges vectorially.
16.8 Electric Field Lines
Electric field lines visually represent the direction and strength of the electric field. They start on positive charges and end on negative charges. The density of lines indicates the field’s strength.
Field lines: Direction is tangent to the field at any point.
Magnitude: Proportional to the density of lines.
Electric dipole: Two equal and opposite charges.
Uniform field: Between parallel plates, the field is constant.
16.9 Electric Fields and Conductors
In electrostatic equilibrium, the electric field inside a conductor is zero. Any excess charge resides on the surface, and the electric field at the surface is perpendicular to it.
Field inside conductor: Zero in static conditions.
Surface charge: Net charge is on the surface.
Field direction: Perpendicular to the surface.
16.10 Electric Forces in Molecular Biology: DNA Structure and Replication
Electrostatic forces play a crucial role in molecular biology, particularly in the structure and replication of DNA. The double helix structure of DNA is stabilized by electrostatic attractions between nucleotide bases (A-T and G-C pairs).
DNA: Double helix structure.
Base pairing: A-T and G-C pairs attract via electrostatic forces.
Replication: Correct bases are attracted during DNA synthesis.
16.11 Photocopy Machines and Computer Printers Use Electrostatics
Electrostatics is used in photocopy machines and laser printers. In these devices, patterns of electric charge are used to attract toner particles to paper, forming images and text.
Photocopying: Uses static charge to transfer toner.
Laser printing: Computer controls laser to create charge patterns.
16.12 Gauss’s Law
Gauss’s law relates the electric flux through a closed surface to the net charge enclosed by that surface. Electric flux is proportional to the number of electric field lines passing through an area.
Electric flux:
Flux through a closed surface:
Gauss’s law:
Application: Useful for calculating electric fields in symmetric situations.
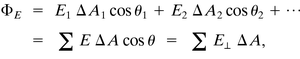
Summary Table: Conductors vs. Insulators
Property | Conductors | Insulators |
|---|---|---|
Charge flow | Free | Almost none |
Examples | Metals | Glass, rubber |
Charging by conduction/induction | Possible | Not possible |
Key Equations
Coulomb’s Law:
Electric Field (point charge):
Force in Electric Field:
Gauss’s Law:
