 Back
BackElectric Charge and Electrostatics: Foundations and Applications
Study Guide - Smart Notes

Structure of Matter and Fundamental Forces
Overview of the Universe's Structure
The universe is composed of matter and governed by four fundamental forces that dictate the interactions between particles and objects. Understanding these forces is essential for explaining a wide range of physical phenomena, from atomic structure to planetary motion.
Gravity: The force of attraction between masses, responsible for the structure of the universe at large scales (e.g., planetary orbits).
Electromagnetism: The force between charged particles, governing electricity, magnetism, and light.
Strong Force: The force that holds protons and neutrons together in the nucleus.
Weak Force: Responsible for certain types of radioactive decay and nuclear reactions.



Electrostatics: Electric Charge and Interactions
Electrostatic Interactions
Electrostatics deals with the forces and phenomena associated with stationary electric charges. Everyday examples include static electricity and lightning.
Electrostatic interaction: The force between objects due to their electric charges.
Examples: Hair standing on end after rubbing with a balloon, lightning during storms.

Triboelectric Effect and Static Electricity
Rubbing different materials can transfer electrons, causing one object to become positively charged and the other negatively charged. This is known as the triboelectric effect.
Triboelectric series: A list of materials ordered by their tendency to gain or lose electrons.
Demo: Rubbing a glass rod with silk or amber with fur demonstrates static electricity.
Material | Tendency |
|---|---|
Glass | Positive |
Silk | Negative |
Amber | Negative |
Fur | Positive |
Others | See full triboelectric series |

Electric Charge: Properties and Conservation
Electric charge is a fundamental property of matter, existing in two types: positive and negative. Like charges repel, and unlike charges attract.
Electrically neutral: Objects with equal amounts of positive and negative charge.
Net charge: The algebraic sum of all charges in a system.
Conservation of charge: The total charge in a closed system remains constant.

Quantization of Charge
Electric charge is quantized, meaning it exists in discrete amounts. The smallest unit of charge is the elementary charge, carried by a proton or electron.
Quantization formula:
where
Elementary charge: C
Coulomb (C): The SI unit of charge. One coulomb is the charge transferred by a current of one ampere in one second.

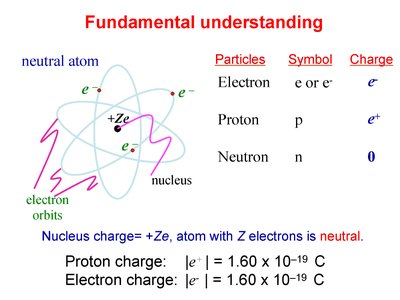
Atomic Structure and Charge
Atoms consist of a nucleus (protons and neutrons) surrounded by electrons. The charges of these particles determine the overall charge of the atom.
Particle | Symbol | Charge |
|---|---|---|
Electron | e or e- | -e |
Proton | p | +e |
Neutron | n | 0 |
Proton charge: C
Electron charge: C
Conductors, Insulators, and Related Phenomena
Classification of Materials
Materials are classified based on their ability to allow electric charges to move.
Conductors: Allow charges to move freely (e.g., metals, tap water, the human body).
Insulators (Dielectrics): Charges are fixed in place (e.g., air, glass, plastic).
Semi-conductors: Intermediate behavior (e.g., silicon, germanium).
Super-conductors: Exhibit zero resistance to charge movement.

Charge Redistribution in Conductors
When a charged object is brought near a conductor, free electrons redistribute in response to the external charge, leading to phenomena such as induction.
Electrons move away from a negatively charged rod, leaving a positive region.
Induced charges can be observed in experiments with metal balls and rods.

Electroscope: Detecting Electric Charge
An electroscope is a device used to detect the presence and magnitude of electric charge. The angle of deflection of its needle or leaves indicates the amount of charge.
Contact with a charged object causes the leaves to repel due to like charges.
The greater the charge, the larger the deflection.

Polarization of Insulators
Insulators do not have free electrons, but their atoms can become polarized in the presence of an external electric field, causing a slight shift of positive and negative charges within the atoms or molecules.
Polarization leads to weak attraction between charged objects and neutral insulators.
Demonstrated by the alignment of molecules in plastic or glass when near a charged rod.

Polarization of Conductors and Insulators
Both conductors and insulators can be polarized, but the mechanisms differ. In conductors, free electrons move, while in insulators, only slight shifts within atoms or molecules occur.
Charge separation is more pronounced in conductors.
In insulators, polarization does not result in net charge separation.

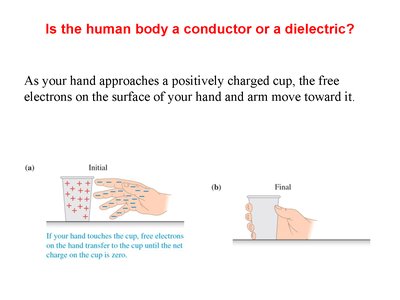
The Human Body: Conductor or Dielectric?
The human body acts as a conductor because it contains free ions and water, allowing electrons to move in response to external electric fields.
When a hand approaches a charged object, electrons move toward or away from the surface, depending on the charge.
Grounding
Grounding provides a path for electric charge to move between an object and the Earth, neutralizing excess charge. This is a common safety practice in electrical systems.
If an object is positively charged, electrons flow from the ground to neutralize it.
Grounding prevents the buildup of dangerous static charges.

Additional info:
These notes cover foundational concepts in electrostatics, relevant to introductory college physics courses and aligned with topics such as electric charge, conductors and insulators, and polarization.
