 Back
BackElectric Charge, Charging Methods, and Coulomb’s Law
Study Guide - Smart Notes

Electric Charge
Definition and Properties
Electric charge is a fundamental property of matter that causes objects to experience a force when placed in an electric or magnetic field. The basic unit of charge is the coulomb (C), and the elementary charge is C. Objects are electrically neutral when they have equal numbers of protons and electrons, negatively charged when they have an excess of electrons, and positively charged when they have a deficiency of electrons.
Charged objects exert non-contact forces: like charges repel, unlike charges attract.
Action-at-distance is a key feature of electric forces.
Methods of Charging
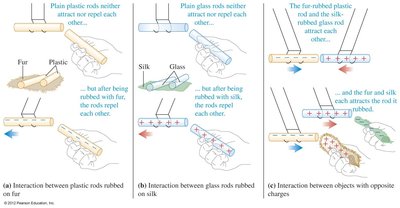
Charging by Friction (Triboelectric Effect)
Charging by friction occurs when two different materials are rubbed together, causing electrons to transfer from one material to another. The triboelectric series ranks materials by their tendency to gain or lose electrons.
Triboelectric effect: Contact electrification due to rubbing.
Materials higher in the triboelectric series lose electrons more easily and become positively charged.
Materials lower in the series gain electrons and become negatively charged.
Material | Charge Tendency |
|---|---|
Glass, Mica, Nylon, Fur, Silk | Positive (loses electrons) |
Paper, Cotton, Wood, Acrylic | Intermediate |
Polystyrene, Rubber, Sulfur, Saran Wrap | Negative (gains electrons) |

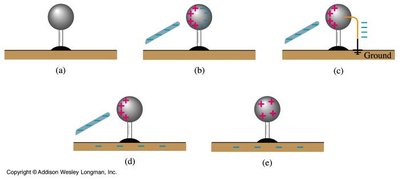
Charging by Induction
Charging by induction is a process where a charged object induces a charge in another object without direct contact. This method relies on the movement of electrons in conductors.
Conductors: Materials where electrons can move freely (e.g., metals).
Insulators (Dielectrics): Materials where electrons are not free to move (e.g., glass, rubber).
Induction involves bringing a charged object near a conductor, causing electrons to redistribute, and then grounding the conductor to allow charge transfer.
Polarization
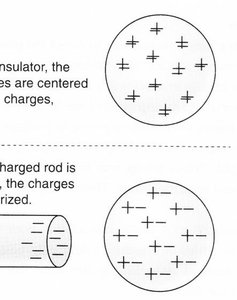
Polarization occurs when the charges within an insulator rearrange in response to a nearby charged object, even though the object itself remains electrically neutral. This results in attractive forces between the charged and neutral objects.
Polarization is significant in dielectrics, where electrons shift slightly within atoms or molecules.
Uncharged objects can be attracted to charged objects due to polarization.
Conductors vs. Insulators
Definitions and Examples
Conductors and insulators differ in their ability to allow electric charges to move.
Conductors: Allow free movement of electrons (e.g., copper, gold).
Insulators: Restrict movement of electrons (e.g., wood, glass, rubber).
Coulomb’s Law
Quantifying Electric Forces
Coulomb’s Law describes the force between two point charges. The force is proportional to the product of the charges and inversely proportional to the square of the distance between them.
Formula:
: Charges (in C)
: Distance between charges (in m)
: Permittivity of free space ()
Forces are equal in magnitude and opposite in direction: .
Example Calculation
Assume a proton ( C) is at the origin, and an electron is at (x=2, y=2) m. Calculate:
(a) The magnitude of the electric force the proton exerts on the electron.
(b) The direction (unit vector) of the force.
Solution:
Where m.
Direction:
Summary Table: Methods of Charging
Method | Description | Key Feature |
|---|---|---|
Friction | Transfer of electrons by rubbing | Depends on triboelectric series |
Induction | Redistribution of electrons without contact | Requires conductors |
Polarization | Rearrangement of charges in insulators | Causes attraction to charged objects |
Applications and Examples
NASA’s triboelectrification rule: Launches may be canceled if vehicles are expected to pass through certain clouds due to charge buildup risks.
Everyday examples: Static electricity, attraction of paper bits to a charged rod.

Additional info: Academic context was added to clarify the triboelectric series, polarization, and the example calculation for Coulomb’s Law.
