 Back
Back1/15 Electric Charge, Coulomb’s Law, and Force Interactions
Study Guide - Smart Notes

Electric Charge and Conservation
Quantization and Conservation of Charge
Electric charge is a fundamental property of matter, quantized in units of the elementary charge e. The unit of charge is the Coulomb (C), where C. Charge is conserved in all physical processes, meaning the total charge in a closed system remains constant.
Atoms are neutral when they have equal numbers of positive (protons) and negative (electrons) charges.
Ions are atoms or molecules with net positive or negative charge.
Conservation of Charge: Charge cannot be created or destroyed, only transferred.
Quantization: Charge exists in discrete units, multiples of .
Interaction between Charges: Like charges repel, unlike charges attract.
Conductors and Insulators
Properties and Examples
Materials are classified based on their ability to allow charge to move freely.
Conductors: Allow free movement of charge (e.g., metals).
Insulators: Do not allow free movement of charge (e.g., glass, plastic).
Charging of Materials & The Electroscope
Methods of Charging and Electroscope Function
Materials can be charged by friction, contact, or induction. An electroscope is a device used to detect the presence of electric charge.
Charging by Friction: Rubbing materials transfers electrons.
Charging by Contact: Touching a charged object to a neutral one transfers charge.
Charging by Induction: Bringing a charged object near a conductor causes redistribution of charge without direct contact.
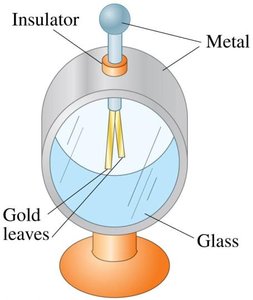
Electroscope: Consists of metal, glass, and gold leaves. When charged, the leaves repel due to like charges.
Coulomb’s Law
Force Between Point Charges
Coulomb’s law describes the force between two point charges. The force can be attractive or repulsive depending on the sign of the charges.
Formula:
Constant: N·m2/C2, , C2/N·m2
Same sign charges: Repel each other.
Opposite sign charges: Attract each other.
Comparison to Gravitational Force
Similarities and Differences
Coulomb’s law and Newton’s law of gravitation both describe inverse-square forces, but differ in their nature and magnitude.
Gravitational Force:
Coulomb Force:
Comparison: Coulomb force can be attractive or repulsive; gravitational force is always attractive.
Magnitude: For protons, Coulomb force is much stronger than gravitational force ( times greater).
Particle | Mass (kg) | Charge (C) |
|---|---|---|
Proton | 1.67 × 10-27 | +1.60 × 10-19 |
Electron | 9.11 × 10-31 | -1.60 × 10-19 |
Force from Multiple Charges
Vector Addition of Forces
The net force on a charge due to multiple other charges is found by vector addition of individual forces.
Superposition Principle: The total force is the sum of forces from each charge.
Example: For charges arranged on a square, forces in the y-direction may cancel, leaving only x-components.
Equilibrium: A charge is in equilibrium when the net force is zero; location depends on the relative magnitudes and positions of other charges.
Charge Density
Uniform Distribution in Spheres
Charge density is the amount of charge per unit volume. For spheres with the same total charge but different sizes, the smallest sphere has the largest charge density.
Charge Density Formula: , where is the volume.
Example: Four spheres with equal charge; the smallest sphere has the highest charge density.

Applications and Example Problems
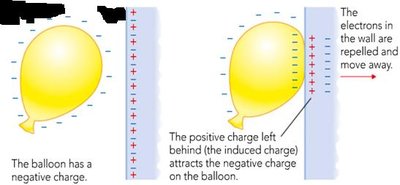
Balloon Demo: Electrostatic Attraction
A negatively charged balloon sticks to a wall due to electrostatic induction. The wall’s electrons are repelled, leaving behind positive charges that attract the balloon.
Can the balloon stay forever? No, eventually the charge dissipates.
Electroscope Example
Charging glass and PVC rods and using an electroscope demonstrates the detection and measurement of electric charge.

Example Problem: Net Force Calculation
Given charges arranged in a square, calculate the net force on a central charge by summing vector components. Forces in the y-direction cancel, leaving only x-components.
Example Problem: Equilibrium Location
To place a third charge in equilibrium between two charges of unequal magnitude, it must be positioned where the forces from both charges balance. The equilibrium location is independent of the third charge’s value.
Homework Questions
Q20.13: A charged sphere released above another identical charged sphere will experience both gravitational and electrostatic forces.
Q20.15: Two metal beads share charge upon contact; the force between them depends on the distribution of charge and their separation.
Summary Table: Comparison of Forces
Force | Formula | Nature | Relative Strength (protons) |
|---|---|---|---|
Electrostatic | Attractive or Repulsive | times stronger than gravity | |
Gravitational | Always Attractive | Much weaker at atomic scale |
Additional info: Academic context was added to clarify the principles of charge conservation, Coulomb’s law, and vector addition of forces, as well as to provide self-contained explanations for example problems and applications.
