 Back
BackElectric Charges and Fields: Structured Study Notes
Study Guide - Smart Notes

Electric Charges and Fields
Introduction to Electrostatics
Electrostatics is the branch of physics that studies forces, fields, and potentials arising from static (non-moving) electric charges. Everyday phenomena such as sparks from synthetic clothes, lightning, and shocks from touching metal objects are explained by the discharge of accumulated static electricity.
Static Electricity: Refers to electric charges at rest.
Electrostatics: The study of static electric charges and their interactions.
Example: Lightning during thunderstorms is a large-scale discharge of static electricity.
Electric Charge
Electric charge is a fundamental property of matter responsible for electric forces. There are two types of charges: positive and negative. Like charges repel, unlike charges attract.
Polarity: The property distinguishing positive and negative charges.
Electrification: Objects acquire charge by rubbing, transferring electrons.
Neutralization: When oppositely charged bodies touch, their charges neutralize.
Convention: Glass rod rubbed with silk becomes positive; silk becomes negative.
Conductors, Insulators, and Semiconductors
Materials are classified based on their ability to allow electric charge movement.
Conductors: Allow free movement of charges (e.g., metals, human body).
Insulators: Resist charge movement (e.g., glass, plastic, wood).
Semiconductors: Intermediate behavior between conductors and insulators.
Example: A metal spoon does not retain charge after rubbing, but a plastic comb does.
Basic Properties of Electric Charge
Electric charge exhibits three fundamental properties: additivity, conservation, and quantization.
Additivity: Total charge is the algebraic sum of individual charges.
Conservation: Total charge in an isolated system remains constant.
Quantization: Charge is always an integer multiple of the elementary charge C.
Example: The charge on a body is , where is an integer.
Coulomb’s Law
Coulomb’s law quantifies the force between two point charges. The force is proportional to the product of the charges and inversely proportional to the square of the distance between them.
Mathematical Form:
Vector Form:
Permittivity of Free Space:
Example: The force between two 1 C charges 1 m apart is N.
Superposition Principle
The force on a charge due to multiple other charges is the vector sum of the individual forces exerted by each charge.
Mathematical Form:
Example: The net force on a charge at the centroid of an equilateral triangle with three equal charges is zero due to symmetry.
Electric Field
An electric field is a region around a charged object where another charge experiences a force. The field is defined as the force per unit positive test charge.
Mathematical Form:
Force Relation:
Unit: N/C (Newton per Coulomb)
Example: The electric field due to a point charge is radially outward for positive charge, inward for negative charge.
Electric Field Due to Multiple Charges
The electric field at a point due to a system of charges is the vector sum of the fields due to each charge.
Mathematical Form:
Electric Field Lines
Electric field lines are visual representations of the direction and strength of the electric field. They start from positive charges and end at negative charges.
Properties:
Field lines never cross.
Density of lines indicates field strength.
Field lines are continuous and do not form closed loops.
Example: Field lines around a dipole show attraction between opposite charges.
Electric Flux
Electric flux measures the number of electric field lines passing through a surface. It is proportional to the strength of the field and the area perpendicular to the field.
Mathematical Form:
Total Flux:
Unit: N m2 C-1
Electric Dipole
An electric dipole consists of two equal and opposite charges separated by a distance. The dipole moment is a vector pointing from negative to positive charge.
Dipole Moment:
Field on Axis:
Field on Equatorial Plane:
Example: Water molecules have a permanent dipole moment.
Dipole in a Uniform External Field
A dipole in a uniform electric field experiences a torque that tends to align it with the field, but no net force.
Torque:
Example: A charged comb polarizes a piece of paper, causing attraction.
Continuous Charge Distribution
Charge can be distributed over a line, surface, or volume. The charge density describes how charge is spread.
Linear Charge Density: (C/m)
Surface Charge Density: (C/m2)
Volume Charge Density: (C/m3)
Gauss’s Law
Gauss’s law relates the electric flux through a closed surface to the total charge enclosed by the surface.
Mathematical Form:
Gaussian Surface: The closed surface chosen for applying Gauss’s law.
Example: The flux through a sphere enclosing a point charge is .
Applications of Gauss’s Law
Gauss’s law simplifies the calculation of electric fields for symmetric charge distributions.
Infinitely Long Wire:
Infinite Plane Sheet:
Thin Spherical Shell:
Outside:
Inside:
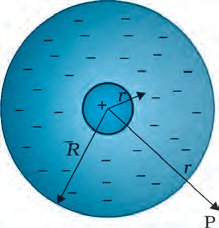
Example: Atom Model with Uniform Charge Distribution
In a model atom with a point nucleus and uniform negative charge distribution:
Charge Density:
Field Inside: for
Field Outside: for
Summary Table: Key Physical Quantities
Physical Quantity | Symbol | Unit | Remarks |
|---|---|---|---|
Vector area element | ΔS | m2 | ΔS = ΔS n̂ |
Electric field | E | V/m | Force per unit charge |
Electric flux | φ | V·m | φ = E·ΔS |
Dipole moment | p | C·m | Vector from -q to +q |
Linear charge density | λ | C/m | Charge per unit length |
Surface charge density | σ | C/m2 | Charge per unit area |
Volume charge density | ρ | C/m3 | Charge per unit volume |

Points to Ponder
Protons are held together in the nucleus by the strong force, not by electric forces.
Coulomb force can be attractive or repulsive, unlike gravity.
Charge is a scalar and is conserved in isolated systems.
Quantization of charge is a fundamental law of nature.
Superposition principle is not obvious; it excludes multi-body forces.
Electric field due to a charge configuration with zero net charge falls off faster than 1/r2.
