 Back
BackElectric Charges and Forces: Structured Study Notes
Study Guide - Smart Notes

Electric Charges and Forces
Introduction to Electric Charge
Electric charge is a fundamental property of matter that gives rise to electric phenomena. Charges interact via long-range forces, similar to gravity, but are much stronger under typical conditions.
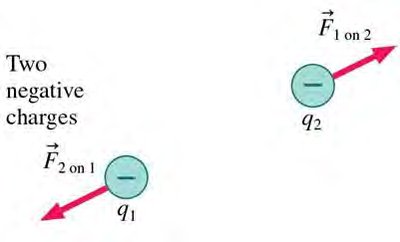
Two Types of Charge: Positive and negative.
Like Charges: Repel each other.
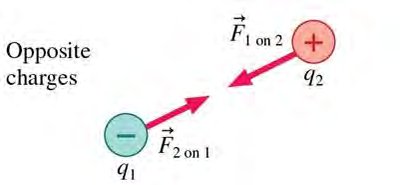
Opposite Charges: Attract each other.
Neutral Objects: Have no net charge.
Charge Conservation: Charge cannot be created or destroyed.
Charge Transfer: Charges can be transferred by contact.
Conductors and Insulators
Materials are classified based on their ability to allow charges to move.
Conductors: Allow charges to move freely (e.g., metals, ionic solutions).
Insulators: Charges are immobile (e.g., glass, plastic).
Atomic Structure and Charge
Atoms consist of a dense nucleus containing protons and neutrons, surrounded by a cloud of electrons. The nucleus is positively charged, while the electron cloud is negatively charged.
Protons: Positive charge, mass ≈ kg, charge C.
Electrons: Negative charge, mass ≈ kg, charge C.
Fundamental Unit of Charge: C.
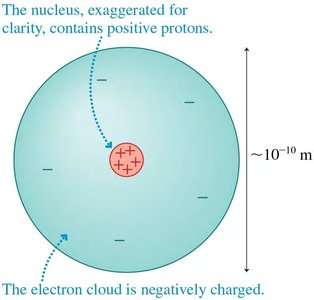
Charge Quantization
Charge is always an integer multiple of the fundamental unit . The net charge of an object is:
Formula:
Neutral Objects: , so .
Charged Objects: .
Polarization and Dipoles
When a charge is brought near an atom, the electron cloud shifts, causing polarization. A separation of equal positive and negative charges forms a dipole.
Polarization: Separation of charge within an atom or molecule.
Dipole: Equal and opposite charges separated by a distance.
Induced Dipole: Created by external electric fields.
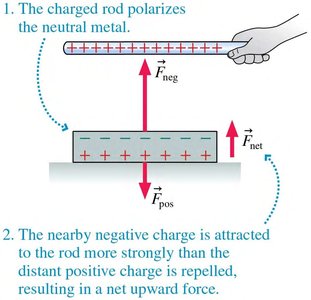
Polarization in Conductors
In conductors, charges shift to the surface, creating a net attractive force toward the external charge.
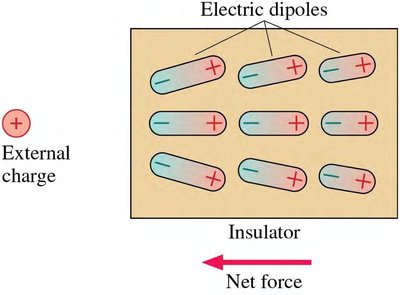
Polarization in Insulators
In insulators, individual atoms become polarized, resulting in a weaker net force toward the external charge.
Charging by Induction
Charging by induction involves bringing a charged object near a conductor, causing charge separation, and then separating the conductors to obtain net charge.
Steps: Bring charged rod near, separate conductors, remove rod.
Result: Net charge on each conductor.

Units of Charge
Charge is measured in Coulombs (C). The fundamental charge is C. Typical charges encountered are in nanocoulombs (nC) or microcoulombs (μC).
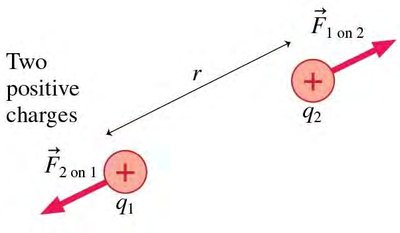
Coulomb’s Law
Coulomb’s law describes the force between two point charges:
Formula:
Electrostatic Constant: N·m2/C2
Direction: Along the line joining the charges; repulsive for like charges, attractive for opposite charges.
Coulomb’s Law in Terms of Permittivity
The constant is related to the permittivity of free space :
Relationship: , C2/N·m2
Alternate Formula:
Coulomb’s Law in Vector Form
The force can be written as a vector:
Formula:
Unit Vector: specifies direction.
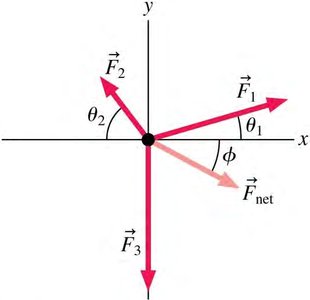
Vector Addition in Electrostatics
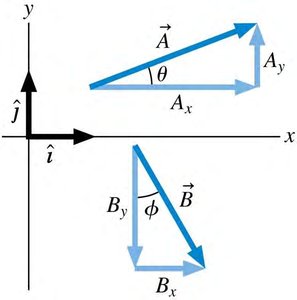
Electric and magnetic forces are vectors and must be added using vector addition. Components are used to simplify calculations.
Vector Components:
Magnitude:
Direction:
Net Force:
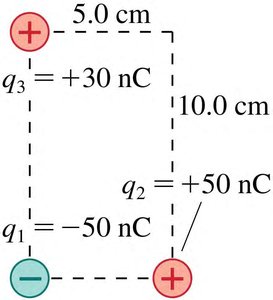
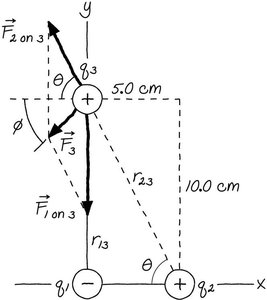
Example: Three Charges
Three point charges are placed at the corners of a rectangle. The net force and electric field at one charge can be calculated using Coulomb’s law and vector addition.
Force Calculation:
Component Form: N
Magnitude: N
Direction:
The Field Model
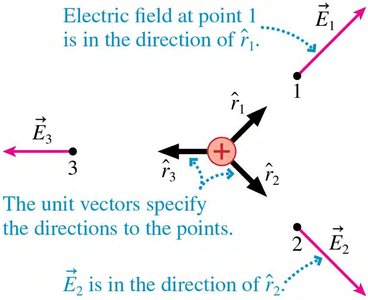
Fields assign a vector to every point in space, representing the influence of charges or masses. The electric field is the alteration of space around a charge.
Electric Field:
Units: N/C
Field Strength: Magnitude of


The Electric Field of a Point Charge
The electric field at a distance from a point charge is:
Formula:
Direction: Away from positive charge, toward negative charge.
Vector Form:
Example: Electric Field at a Point
To find the electric field at the location of a charge due to other charges, sum the fields from each charge using vector addition.
Component Form: N/C
Everyday Applications: Water Molecule
Water is a polar molecule with a dipole moment, making it a good solvent and responsible for properties like surface tension and high heat capacity.
Dipole Moment: C·m
Summary Table: Properties of Protons and Electrons
Particle | Mass (kg) | Charge | Charge (C) |
|---|---|---|---|
Proton | 1.67 × 10−27 | +e | +1.6 × 10−19 |
Electron | 9.11 × 10−31 | −e | −1.6 × 10−19 |
Additional info: Table values inferred and corrected for clarity.
