 Back
BackElectric Fields and Forces: Fundamental Concepts and Applications
Study Guide - Smart Notes

Electric Fields and Forces
Fundamental Forces
The universe is governed by four fundamental forces: gravity, electricity, magnetism, and the strong and weak nuclear forces. These forces are responsible for the interactions between particles and objects at all scales.
Gravity: The force of attraction between masses.
Electricity: The force between charged particles.
Magnetism: The force between magnetic materials.
Strong and Weak Nuclear Forces: Forces acting within atomic nuclei.

Electricity and Magnetism in Nature
Electricity and magnetism have been observed since ancient times. Amber, when rubbed, attracts small objects due to static electricity, while lodestone naturally attracts iron due to magnetism.
Amber: Exhibits static electricity when rubbed.
Lodestone: Naturally magnetic mineral.

20.1 Charges and Forces
Sources of Forces: Mass and Charge
In classical mechanics, mass is the source of gravity. Similarly, electrical charges are the source of electric forces. Objects with mass exert gravitational forces, while objects with charge exert electric forces.
Gravitational Force: Acts between masses.
Electrostatic Force: Acts between charges.
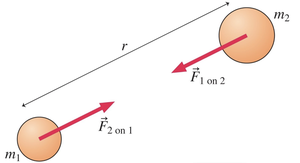
Types of Electric Charge
There are two types of electric charge: positive and negative. Like charges repel, and opposite charges attract. These interactions are called electrostatic forces.
Positive Charge: Denoted by a plus sign (+).
Negative Charge: Denoted by a minus sign (-).
Electrostatic Forces: Attraction or repulsion between charges.
Neutral Objects
Objects without a net charge are neutral and do not experience electrical forces. Neutrality is indicated by the absence of charge symbols in diagrams.
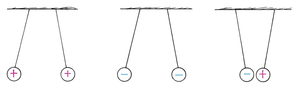
Visualizing Charge: Charge Diagrams
Charge diagrams are schematic representations showing the distribution and net charge on objects. The number of plus or minus signs indicates the amount and type of charge.
Equal amounts of positive and negative charge: Object is neutral.
More charge: Represented by more plus or minus signs.
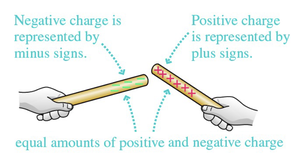

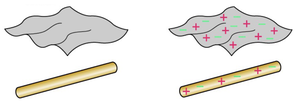
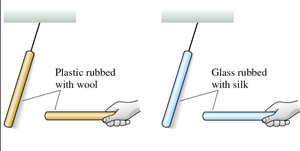
Charging Through Friction
When two objects are rubbed together, charges are transferred from one to the other. This process is called charging by friction. For example, rubbing a plastic rod with wool transfers electrons, making the rod negatively charged and the wool positively charged.
Charge Transfer: Electrons move from one material to another.
Result: One object gains negative charge, the other gains positive charge.
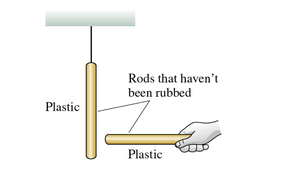
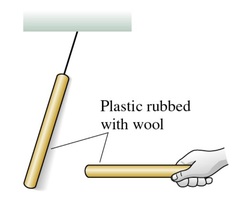
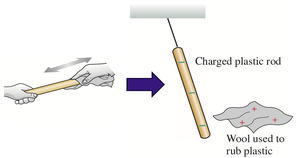
Charging with Different Materials
Rubbing a glass rod with silk results in the glass rod becoming positively charged and the silk negatively charged. The process gives plastic and glass rods opposite charges.
Plastic and Wool: Plastic becomes negative, wool becomes positive.
Glass and Silk: Glass becomes positive, silk becomes negative.
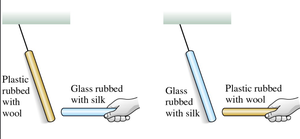
Law of Conservation of Charge
Charge is neither created nor destroyed; it is only transferred. If positive charge appears somewhere, an equal amount of negative charge appears elsewhere, keeping the net charge constant.
Conservation Principle: Total charge remains constant in any process.
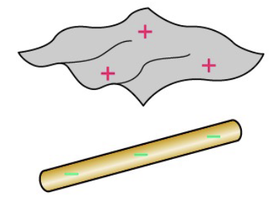
20.2 Charges, Atoms, and Molecules
Atomic Structure and Charge
To understand charging, we must look at the atomic level. Atoms consist of a nucleus containing protons (positive charge) and an electron cloud (negative charge). Electrons are loosely bound and can be removed or added, leading to ionization.
Protons: Positive charge, tightly bound in nucleus.
Electrons: Negative charge, loosely bound, can be transferred.
Ionization: Removal or addition of electrons creates ions.

Charge Conservation and Units
Charge is represented by the symbol q or Q. The SI unit of charge is the Coulomb (C). The fundamental charge e is the magnitude of the charge of an electron or proton: .
Charge Conservation: Electrons and protons are not created or destroyed in ordinary processes.
Positive Ion: Atom missing an electron.
Negative Ion: Atom with an extra electron.
Friction and Molecular Bonds
Friction can break molecular bonds at surfaces, allowing electrons to move from one material to another and create charged materials.
Insulators and Conductors
In conductors (e.g., metals), valence electrons are weakly bound and free to move. In insulators, electrons are tightly bound and not free to move.
Conductors: Allow free movement of electrons.
Insulators: Electrons are not free to move.
Summary Table: Types of Charge and Materials
Type | Charge | Behavior |
|---|---|---|
Proton | +e | Tightly bound in nucleus |
Electron | -e | Loosely bound, can be transferred |
Conductor | Varies | Electrons move freely |
Insulator | Varies | Electrons tightly bound |
