 Back
BackElectrochemistry: Redox Reactions, Batteries, and Electrolysis
Study Guide - Smart Notes

Electrochemistry
Introduction to Electrochemistry
Electrochemistry is the study of the relationship between chemical reactions and electricity. It focuses on the movement of electrons in redox (reduction-oxidation) reactions, which are fundamental to the operation of batteries, electrolysis, and many biological and industrial processes.
Electricity is defined as a flow of electrons.
Electrons in electrochemical systems originate from redox reactions.
Redox reactions involve both oxidation (loss of electrons) and reduction (gain of electrons) occurring simultaneously.
Redox Reactions
Oxidation and Reduction
Redox reactions are split into two half-reactions: oxidation and reduction. Each half-reaction shows either the loss or gain of electrons.
Oxidation: Loss of electrons (e.g., )
Reduction: Gain of electrons (e.g., )
Each half-reaction can be balanced separately before combining to form the overall redox equation.
Assigning Oxidation Numbers
Oxidation numbers help identify which species are oxidized and reduced in a reaction. Common rules include:
Hydrogen: +1 (except in hydrides, where it is -1)
Group 1A: +1; Group 2A: +2
Halogens: -1
Oxygen: -2 (except in peroxides, where it is -1)
Sulfur: -2; Nitrogen and Phosphorus: -3 (if only anion)
Balancing Redox Reactions
Redox reactions are balanced differently in acidic and basic solutions:
Balance all atoms except O and H.
Add to balance O, to balance H (acidic), or (basic).
Add electrons to balance charge.
Multiply half-reactions so electrons cancel, then combine and simplify.
Example (Acidic):
Assign oxidation numbers, split into half-reactions, balance, and combine.
Example (Basic):
Balance as in acidic, then add to both sides to neutralize , forming .
Electrochemical Cells
Voltaic (Galvanic) Cells
Voltaic cells convert chemical energy into electrical energy through spontaneous redox reactions. They consist of two half-cells connected by a salt bridge.
Anode: Site of oxidation (electrons generated)
Cathode: Site of reduction (electrons consumed)
Electrons flow from anode to cathode through an external circuit.

Cell Notation
Cell shorthand notation summarizes the components of an electrochemical cell:
Format: Anode | Anode solution || Cathode solution | Cathode
Example:
Standard Electrode Potentials
The standard reduction potential () measures the tendency of a half-cell to gain electrons under standard conditions (1 M, 1 atm, 25°C). The cell potential () is calculated as:
A positive indicates a spontaneous reaction.
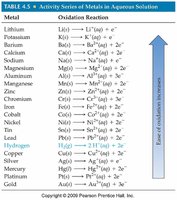
Activity Series of Metals
The activity series ranks metals by their tendency to be oxidized. More active metals are stronger reducing agents.
Metal | Oxidation Reaction |
|---|---|
Lithium | Li(s) → Li+(aq) + e- |
Potassium | K(s) → K+(aq) + e- |
Calcium | Ca(s) → Ca2+(aq) + 2e- |
... | ... |
Gold | Au(s) → Au3+(aq) + 3e- |
Batteries
Common Battery Types and Chemistry
Batteries are practical applications of voltaic cells. Different types use various redox couples and electrolytes.
Dry Cell (Leclanché cell): Uses Zn and MnO2 with a moist paste electrolyte.
Mercury Battery: Zn(Hg) and HgO, used in small devices.
Lead-Acid Battery: Used in cars; consists of Pb, PbO2, and H2SO4.
Nickel-Cadmium (NiCad): Rechargeable, uses Cd and NiO2.
Lithium-Ion Battery: Lightweight, high energy density, uses LiCoO2 and graphite.

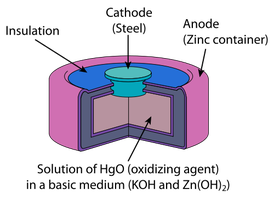
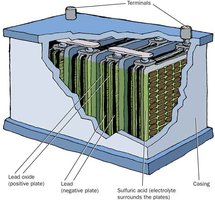






Cell Potential, Free Energy, and Equilibrium
Relationship Between and
The maximum electrical work from a cell is related to the change in Gibbs free energy:
= number of electrons transferred
= Faraday constant ( C/mol e-)
A positive means a negative (spontaneous reaction).
The equilibrium constant is related to by:
(at 25°C)
The Nernst Equation
The Nernst equation allows calculation of cell potential under non-standard conditions:
= reaction quotient (ratio of product and reactant concentrations)
Measuring pH with Electrochemistry
pH Meters and Electrochemical Cells
pH meters use electrochemical cells to measure the voltage difference between a test solution and a reference, which is related to the hydrogen ion concentration.

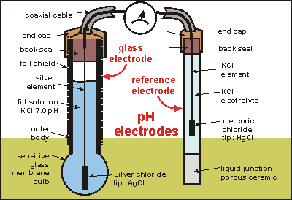
Electrolysis
Principles of Electrolysis
Electrolysis uses electrical energy to drive non-spontaneous chemical reactions. It is the reverse of a voltaic cell and requires an external power source.
Used for metal extraction, purification, and electroplating.
Common products: Li, Na, Ca, K, Mg, Al, Cl2, NaOH, H2.
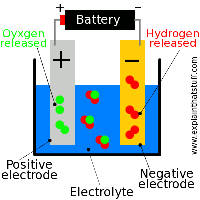
Electrolysis of Molten and Aqueous Salts
Electrolysis of molten NaCl produces Na metal and Cl2 gas. In aqueous solutions, water can also be oxidized or reduced, affecting the products.
At the cathode: or
At the anode: or
In practice, industrial conditions favor Cl2 production at the anode.
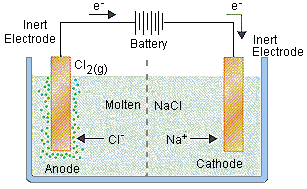
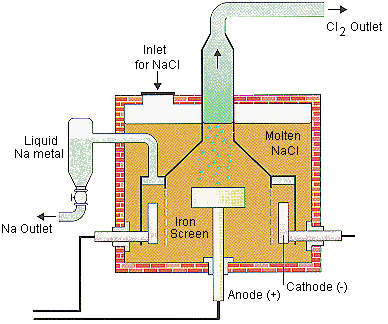
Applications of Electrolysis
Production of metals (e.g., Na, Al)
Electroplating
Production of industrial chemicals (e.g., Cl2, NaOH)
Oxygen generation in spacecraft and submarines
Additional info: This summary expands on the provided notes with definitions, examples, and equations for clarity and completeness, suitable for college-level physics and chemistry students.
