 Back
BackElectrostatics and Charge: Study Guide for College Physics
Study Guide - Smart Notes

Electrostatics
Introduction to Electrostatics
Electrostatics is the study of stationary electric charges and the forces they exert. It forms the foundation for understanding electric fields, forces, and the behavior of charged particles.
Subatomic Particles and Charge
Types of Subatomic Particles
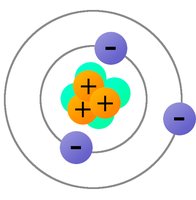
Atoms are composed of protons (p+), electrons (e-), and neutrons. Protons carry a positive charge, electrons carry a negative charge, and neutrons are neutral. The arrangement of these particles determines the overall charge of an object.
Protons (p+): Positively charged particles found in the nucleus.
Electrons (e-): Negatively charged particles orbiting the nucleus.
Neutrons: Neutral particles in the nucleus (not directly involved in electrostatics).
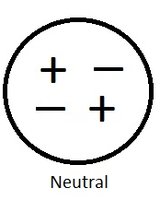
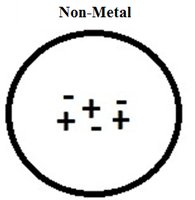
Neutral Objects
An object is neutral when it contains equal numbers of protons and electrons. This balance results in no net charge.
Charging and Transfer of Electrons
Charging by Electron Transfer
Objects can become charged by gaining or losing electrons. The transfer of electrons from one object to another changes their net charge.
Gaining electrons: Results in a negatively charged object.
Losing electrons: Results in a positively charged object.
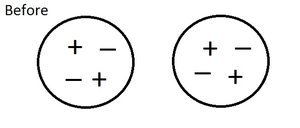
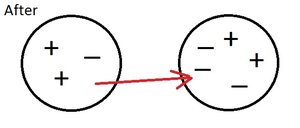
Negatively Charged Objects
When an object has more electrons than protons, it is negatively charged.
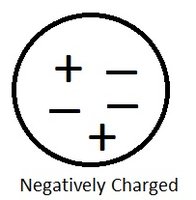
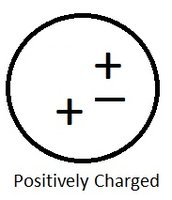
Positively Charged Objects
When an object has fewer electrons than protons, it is positively charged.
Law of Conservation of Charge
Conservation Principle
The Law of Conservation of Charge states that the total charge in a closed system remains constant. When electrons are transferred, the loss in one object equals the gain in another.
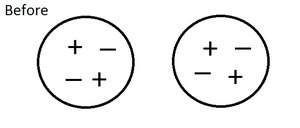
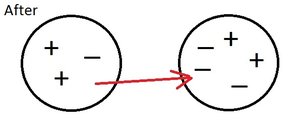
Electrostatic Forces and Fields
Electrostatic Field
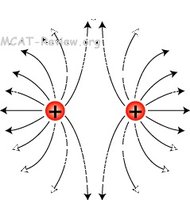
An electrostatic field is the region around a charged object where electric forces are exerted on other charges. Field lines indicate the direction and strength of the force.
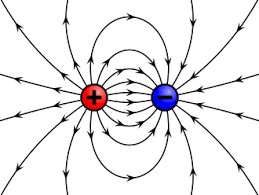
Field Direction
Field lines always point from positively charged objects to negatively charged objects. The pattern of field lines shows attraction or repulsion between charges.
Opposite charges: Attract each other.
Like charges: Repel each other.
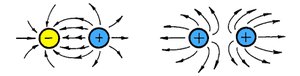
Methods of Charging
Charging by Friction
When two objects are rubbed together, electrons may transfer from one to the other. The object that loses electrons becomes positively charged, and the one that gains electrons becomes negatively charged.
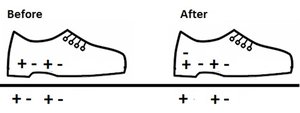
Charging by Conduction
Charging by conduction occurs when a charged object touches another object, transferring electrons directly.
Conductors and Insulators
Conductors
Conductors are materials, typically metals, that allow free movement of electrons. They can be easily charged and conduct electricity.
Example: Copper wire

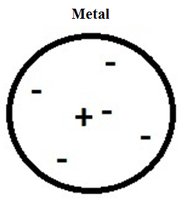
Insulators
Insulators are materials, usually non-metals, with tightly held electrons that do not move freely. They are poor conductors of electricity.
Example: Wood

Grounding
Ground
A ground is an object large enough to gain or lose electrons without becoming noticeably charged. It serves as a reservoir for charge.

Electrostatic Field Patterns
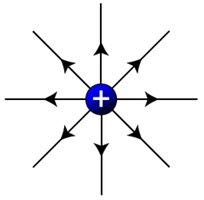
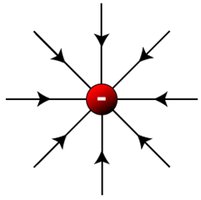
Field Lines for Different Charges
Field lines illustrate the direction of force exerted by charges:
Positive charge: Field lines point outward.
Negative charge: Field lines point inward.
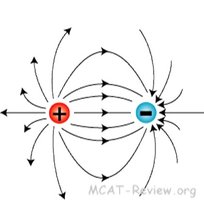
Interaction of Charges
When opposite charges are near each other, field lines connect them, showing attraction. Like charges have field lines that repel.
Charge and Force Relationships
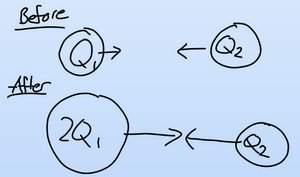
Direct Proportionality
The force between two charged objects increases as the magnitude of their charges increases. This relationship is directly proportional.
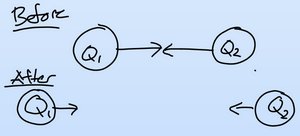
Inverse Square Law
The force between two charges decreases as the distance between them increases, following an inverse square law.
Coulomb's Law
Formula and Explanation
Coulomb's Law quantifies the force between two point charges:
k: Coulomb's constant ( Nm2/C2)
q1, q2: Charges (Coulombs)
r: Distance between charges (meters)
The law states:
The force is directly proportional to the product of the charges and inversely proportional to the square of the distance between them.
Example Calculation
Two objects each have a charge of 20.0 C. If the distance between them is 0.5 m, what is the force?
Calculate the value to find the force in Newtons.
Summary Table: Charge Types and Properties
Type | Charge | Behavior |
|---|---|---|
Neutral | Equal protons and electrons | No net charge |
Positive | More protons than electrons | Repels other positive, attracts negative |
Negative | More electrons than protons | Repels other negative, attracts positive |
