 Back
BackFluids: Properties, Pressure, and Dynamics
Study Guide - Smart Notes

9.1 Properties of Fluids
Definition and Types of Fluids
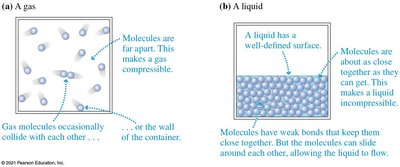
Fluids are substances that can flow and take the shape of their container. Both liquids and gases are classified as fluids, but they have distinct molecular arrangements and properties.
Gases: Molecules are far apart, making gases compressible. Gas molecules move freely and occasionally collide with each other or the walls of the container.
Liquids: Molecules are close together, making liquids essentially incompressible. Molecules have weak bonds but can slide past each other, allowing flow.
Volume and Density
Volume is the amount of space a system occupies, measured in cubic meters (m3). Density (\( \rho \)) is the ratio of mass to volume:
SI units for density are kg/m3, but g/cm3 is also common.
9.2 Pressure in Fluids
Definition of Pressure
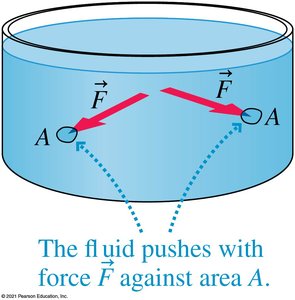
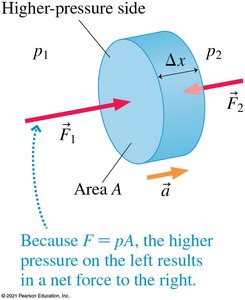
Pressure is the ratio of force to the area over which the force is exerted:
Pressure is a scalar quantity and is measured in pascals (Pa), where 1 Pa = 1 N/m2.
Pressure in Liquids and Gases
In liquids, pressure increases with depth due to gravity.
In gases, pressure is nearly uniform in small containers but decreases with height in the atmosphere.
Causes of Pressure
In a weightless environment, liquids do not exert pressure on container walls, but gases do due to molecular collisions.
On Earth, gravity causes liquids to exert pressure on the bottom and sides of containers, and causes a slight density gradient in gases.
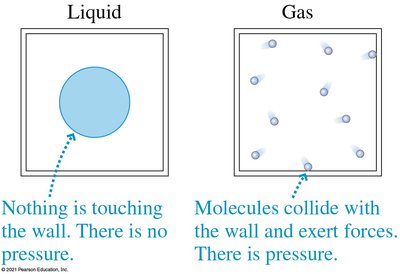
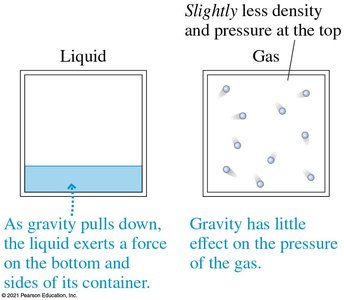
Atmospheric Pressure
Atmospheric pressure decreases with altitude. At sea level, the standard atmospheric pressure is:
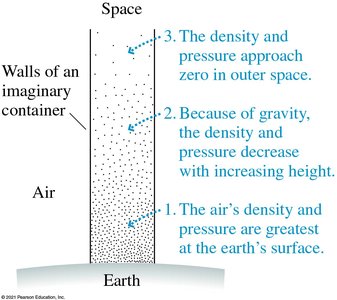
Pressure Forces in Fluids
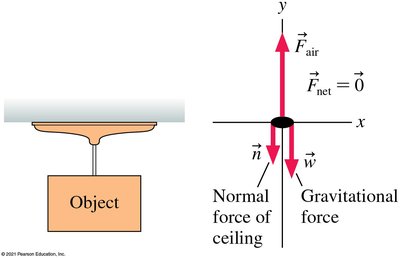
Fluids exert pressure in all directions, resulting in balanced forces on submerged objects. Devices like suction cups work by creating a pressure difference.
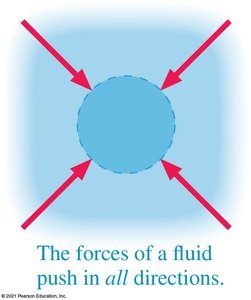
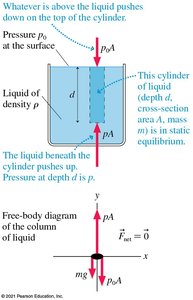
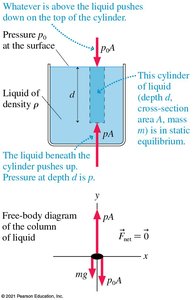
Pressure in Liquids: Hydrostatic Pressure
The pressure at depth d in a liquid is given by:
where is the pressure at the surface, is the liquid's density, and is the acceleration due to gravity.
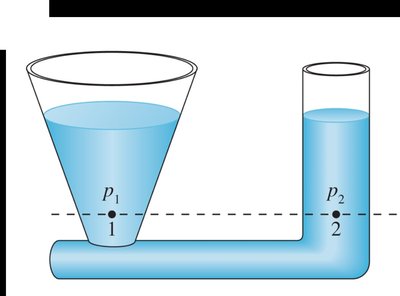
Hydrostatic Equilibrium
In a connected liquid at rest, the pressure is the same at all points on a horizontal line.
Liquids rise to the same height in all open regions of a container.
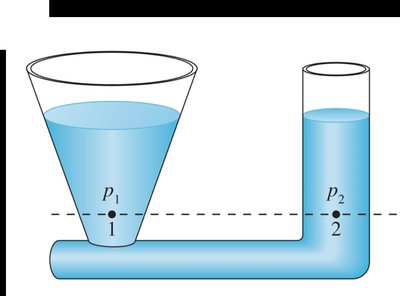
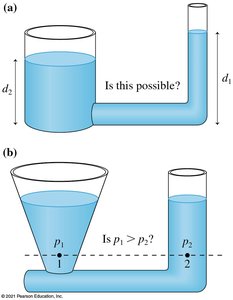
Gauge Pressure and Barometers
Gauge pressure is the pressure in excess of atmospheric pressure. Many devices, such as tire gauges, measure gauge pressure. To find absolute pressure, add atmospheric pressure to the gauge reading:

Barometers measure atmospheric pressure using the height of a liquid column:
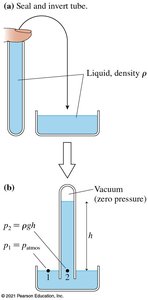
9.3 Buoyancy and Archimedes' Principle
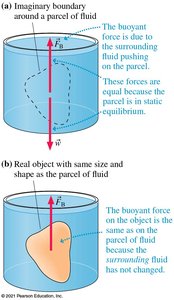
Buoyant Force
Fluids exert an upward force on submerged objects called the buoyant force. This force arises because pressure increases with depth, so the bottom of an object experiences more pressure than the top.
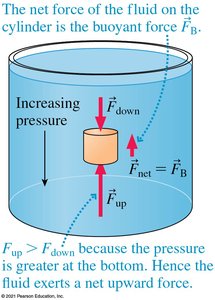
Archimedes' Principle
Archimedes' principle states that the buoyant force on an object is equal to the weight of the fluid displaced by the object:
where is the density of the fluid and is the volume of fluid displaced.
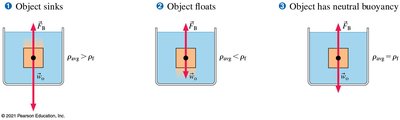
Applications: Floating and Sinking
An object sinks if its average density is greater than the fluid's density.
An object floats if its average density is less than the fluid's density.
Neutral buoyancy occurs when the object's average density equals the fluid's density.
9.5 Fluids in Motion
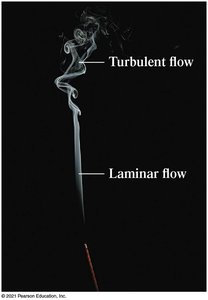
Laminar and Turbulent Flow
Fluid flow can be laminar (smooth and steady) or turbulent (chaotic and irregular). Most introductory analysis assumes laminar flow and incompressible fluids.
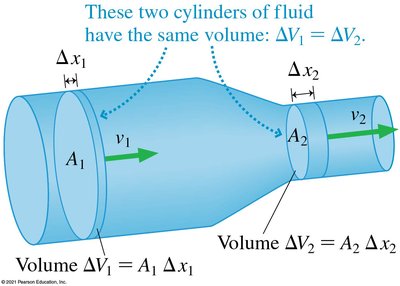
Equation of Continuity
For an incompressible fluid, the volume flow rate is constant throughout a tube:
where is the fluid speed and is the cross-sectional area. The volume flow rate is:

9.6 Ideal Fluid Dynamics and Bernoulli's Equation
Bernoulli's Equation
Bernoulli's equation relates pressure and velocity in an ideal (nonviscous, incompressible) fluid:
Pressure is higher where the fluid moves slower, and lower where it moves faster.
9.7 Viscous Fluid Dynamics
Viscosity and Poiseuille's Equation
Viscosity (η) is a fluid's resistance to flow. For viscous fluids, a pressure difference is required to maintain flow. The volume flow rate for a viscous fluid in a tube is given by Poiseuille's equation:
where is the tube radius, is the pressure difference, is the tube length, and is the viscosity.
Turbulence and Reynolds Number
Flow becomes turbulent when the Reynolds number (ratio of inertial to viscous forces) exceeds a critical value. Turbulent flow is characterized by chaotic changes in pressure and velocity.
Summary Table: Pressure Units
Unit | Abbreviation | Conversion to Pa | Uses |
|---|---|---|---|
pascal | Pa | 1 Pa = 1 N/m2 | SI unit, most calculations |
atmosphere | atm | 1 atm = 101 kPa | General |
millimeters of mercury | mm Hg | 1 mm Hg = 133 Pa | Gases, blood pressure, barometric pressure |
pounds per square inch | psi | 1 psi = 6.89 kPa | U.S. engineering and industry |
