 Back
BackFoundations of Modern Physics: Atomic Structure, Spectra, and the Limits of Classical Physics
Study Guide - Smart Notes

Chapter 37: The Foundations of Modern Physics
Introduction
This chapter explores the experimental discoveries and theoretical developments that led to the modern understanding of atomic structure and the limitations of classical physics. It covers the nature of atomic spectra, the discovery of subatomic particles, and the models of the atom that emerged in the early 20th century.
Atomic Spectra and Blackbody Radiation
Spectroscopy and the Spectrometer
Spectroscopy is the study of the interaction between matter and electromagnetic radiation. A spectrometer is a key instrument for measuring the wavelengths of light, often using a diffraction grating to separate light into its component wavelengths.
Diffraction gratings disperse light into a spectrum, allowing precise measurement of wavelengths.
Curved gratings focus the spectrum onto a photodetector for analysis.
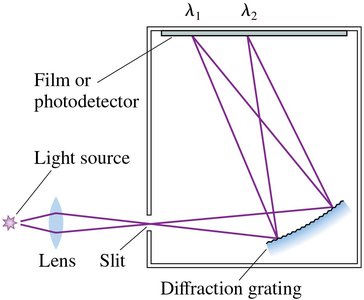
Continuous and Discrete Spectra
Light emitted by different sources can form either a continuous or a discrete spectrum:
Continuous spectrum: Produced by hot, self-luminous objects (e.g., incandescent bulbs), emitting light at all wavelengths.
Discrete spectrum: Produced by gases in discharge tubes, emitting light at specific, unique wavelengths for each element.
Blackbody Radiation
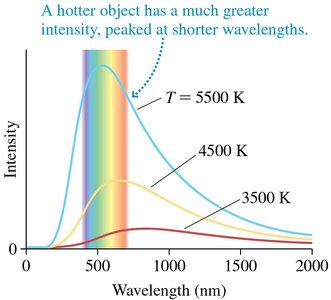
A blackbody is an idealized object that absorbs and emits all frequencies of electromagnetic radiation. The spectrum of blackbody radiation depends only on the object's temperature.
The rate of heat energy emission is given by the Stefan-Boltzmann law: where is emissivity, , is surface area, and is absolute temperature.
Wien's Law gives the wavelength of peak intensity:
Increasing temperature increases intensity and shifts the peak to shorter wavelengths.

Atomic Emission and Absorption Spectra
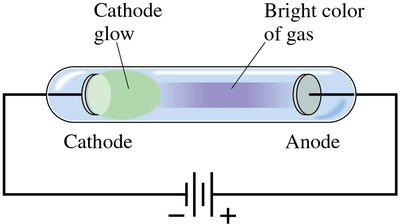
Gas Discharge Tubes and Discrete Spectra
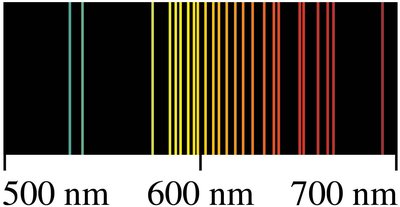
When a high voltage is applied to a gas at low pressure, the gas emits light with a characteristic color. Passing this light through a spectrometer reveals a discrete spectrum unique to each element.
Each element's spectrum acts as a 'fingerprint' for identification.

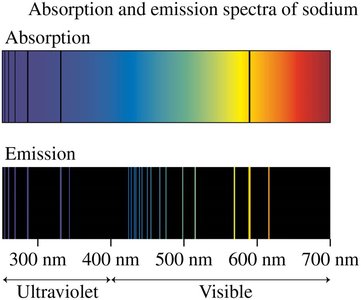
Absorption Spectra
When white light passes through a gas, certain wavelengths are absorbed, resulting in dark lines in the spectrum at those wavelengths. The absorption spectrum is a subset of the emission spectrum for that gas.
Every absorbed wavelength is also emitted, but not all emitted wavelengths are absorbed.
The Hydrogen Emission Spectrum and the Balmer Formula
The hydrogen atom's emission spectrum is simple and regular. Johann Balmer discovered an empirical formula for its wavelengths:
Balmer formula: where (for visible lines), are integers.

Discovery of Subatomic Particles
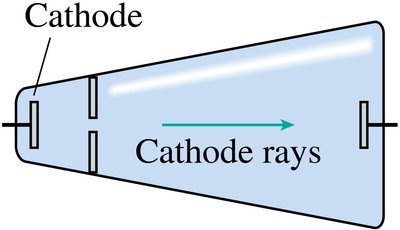
Cathode Rays and the Electron
Experiments with cathode ray tubes revealed the existence of negatively charged particles (electrons) within atoms. J. J. Thomson showed that cathode rays are streams of electrons, which are fundamental constituents of all atoms.
Cathode rays are deflected by magnetic and electric fields, indicating they are charged particles.
Properties of cathode rays are independent of the cathode material.
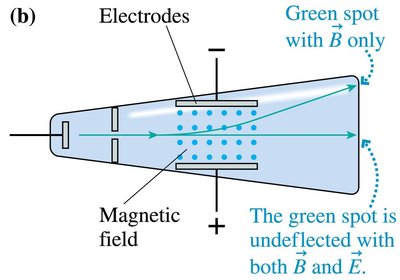
Thomson’s Crossed-Field Experiment
Thomson used electric and magnetic fields to measure the charge-to-mass ratio of the electron. By balancing the forces, he determined:
For a particle to pass undeflected:
Charge-to-mass ratio:
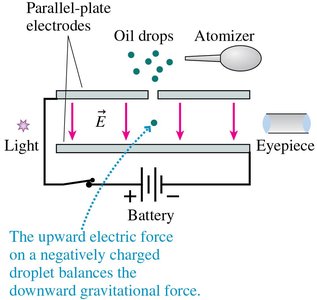
Millikan Oil-Drop Experiment and the Fundamental Charge
Robert Millikan measured the elementary charge by balancing the gravitational and electric forces on tiny charged oil droplets:
Balance condition:
Fundamental charge:
Models of the Atom
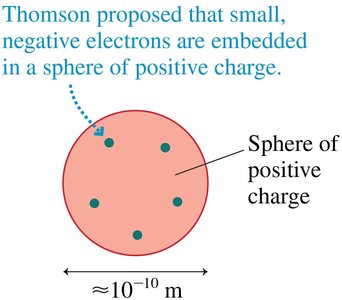
Thomson’s Raisin-Cake (Plum Pudding) Model
Thomson proposed that electrons are embedded in a sphere of positive charge. This model could not explain all experimental results.
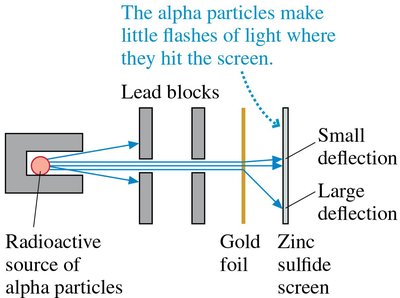
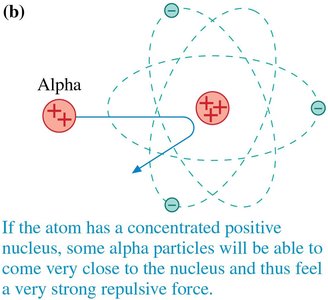
Rutherford’s Gold Foil Experiment and Nuclear Model
Rutherford’s experiment involved firing alpha particles at thin gold foil. Most passed through, but some were deflected at large angles, indicating a small, dense, positively charged nucleus.
Led to the nuclear model: electrons orbit a tiny nucleus.
Most of the atom is empty space.
Structure of the Nucleus
Protons and Neutrons
The nucleus contains protons (positive charge) and neutrons (neutral). The number of protons (atomic number ) defines the element, while the number of neutrons can vary, leading to isotopes.
Mass number (protons + neutrons).
Isotopes: atoms with the same but different .
Limits of Classical Physics
Problems with Classical Models
Classical physics could not explain several phenomena:
Stability of atoms (orbiting electrons should radiate energy and collapse into the nucleus).
Discrete atomic spectra (classical theory predicts continuous spectra).
Blackbody radiation (classical theory predicts the 'ultraviolet catastrophe').
These failures led to the development of quantum theory, which is addressed in subsequent chapters.
Key Equations and Constants
Stefan-Boltzmann Law:
Wien’s Law:
Balmer Formula (Hydrogen):
Elementary charge:
Electron volt:
Summary Table: Atomic Models and Key Discoveries
Model/Discovery | Main Features | Key Experiment |
|---|---|---|
Thomson's Model | Electrons in a sphere of positive charge | Cathode ray tube |
Rutherford's Model | Electrons orbit a dense nucleus | Gold foil experiment |
Electron Discovery | Negative subatomic particle | Cathode rays, crossed-field experiment |
Proton Discovery | Positive charge in nucleus | Scattering experiments |
Neutron Discovery | Neutral particle in nucleus | Mass spectrometry, nuclear reactions |
