 Back
BackFoundations of Physics: Scientific Method, Symmetry, Matter, Units, and Measurement
Study Guide - Smart Notes

Chapter 1: Foundations
1.1 The Scientific Method
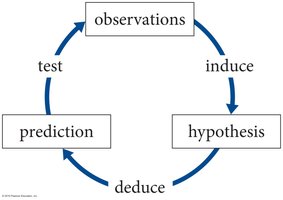
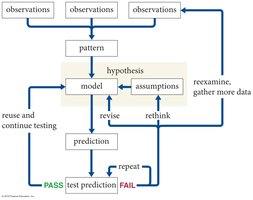
The scientific method is a systematic, iterative process used to develop and test explanations for natural phenomena. It is central to the advancement of physics and all sciences.
Observation: Careful examination of phenomena or experimental results.
Hypothesis: A proposed explanation based on observations, which can be tested.
Prediction: Logical consequences derived from the hypothesis, which can be checked by further observations or experiments.
Testing: Conducting experiments or gathering data to evaluate the prediction.
Iteration: The process is repeated, refining hypotheses and models as new data is collected.
Example: Observing a rock falling leads to the hypothesis that a force pulls objects downward. Experiments can test this hypothesis, and predictions can be made about other objects.
Law vs. Theory:
Law: Describes what happens under certain conditions (e.g., Newton's Laws).
Theory: Explains why something happens, based on underlying principles (e.g., Theory of Relativity).
Science is always evolving; no law or theory is considered absolutely true, and all are subject to revision with new evidence.
1.2 Symmetry
Symmetry is a fundamental concept in physics, referring to properties of objects or laws that remain unchanged under certain operations or transformations.
Translational Symmetry: The laws of physics are the same everywhere in space.
Rotational Symmetry: Physical laws do not change when systems are rotated.
Reflection Symmetry: Systems remain unchanged when reflected across a plane.
Other Symmetries: Charge symmetry, parity symmetry, and time symmetry are examples of more abstract symmetries in physics.
Example: A chessboard pattern that looks the same when shifted demonstrates translational symmetry.
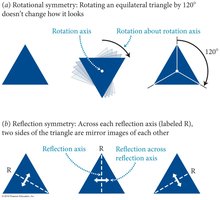

Symmetry is essential for formulating physical laws; if an experiment's outcome is unchanged by a transformation, the law describing it must also possess that symmetry.

Additional info: The search for new symmetries continues to be a major focus in modern physics, as symmetries often lead to conservation laws (e.g., conservation of momentum, energy).
1.3 Matter and the Universe
The universe consists of matter and energy, existing within the framework of space and time. Physicists use measurable quantities to describe and understand the universe.
Matter: Composed of atoms, which consist of protons, neutrons, and electrons.
Energy: The capacity to do work or cause change.
Space and Time: The arena in which all events occur.
Physical Quantities: Properties such as length, mass, and time, which can be measured and expressed with units.

Scale of the Universe: Objects in the universe range from subatomic particles (10-15 m) to galaxies (1021 m).
1.4 Time and Change
Time is a fundamental dimension in physics, providing a framework for the sequence and causality of events.
Arrow of Time: Time flows in one direction, from past to future, establishing causality (cause precedes effect).
Standard Unit: The second (s) is the SI unit of time, defined by the vibrations of cesium atoms.
Principle of Causality: For any two events, if event A causes event B, all observers agree that A occurs before B.
1.5 Representations
Visual representations are crucial tools in physics for organizing information, understanding relationships, and solving problems.
Types: Diagrams, sketches, graphs, and models.
Purpose: Help clarify complex systems, visualize changes, and communicate ideas effectively.
1.6 Physical Quantities and Units
Physical quantities are expressed as the product of a numerical value and a unit. The International System of Units (SI) provides standard units for all physical measurements.
Base SI Units: Meter (m) for length, kilogram (kg) for mass, second (s) for time, ampere (A) for electric current, kelvin (K) for temperature, mole (mol) for amount of substance, candela (cd) for luminous intensity.
Derived Units: Formed by combining base units (e.g., m/s for velocity).
Metric Prefixes: Used to express very large or small quantities (e.g., kilo-, milli-, micro-).
Example:
1.7 Significant Digits
Significant digits (or figures) indicate the precision of a measured or calculated quantity.
Precision: Consistency of repeated measurements.
Accuracy: Closeness to the true value.
Rules:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros after a decimal point are significant.
Use scientific notation to clarify significant digits in ambiguous cases.
1.8 Density and the Mole
Density is a measure of how much of a substance exists in a given volume. The mole is the SI unit for the amount of substance, defined by Avogadro's number.
Number Density: , where is the number of objects and is the volume.
Mass Density: , where is mass and is volume.
Mole (mol): entities (Avogadro's number).
1.9 Unit Conversions
Unit conversions are performed using ratios that equal one, ensuring the value remains unchanged while expressing it in different units.
Example: To convert 4.5 in to mm, use .
Convention: When raising a unit with a prefix to a power, the power applies to both (e.g., ).
Summary Table: SI Base Units
Quantity | Unit | Symbol |
|---|---|---|
Length | meter | m |
Mass | kilogram | kg |
Time | second | s |
Electric current | ampere | A |
Temperature | kelvin | K |
Amount of substance | mole | mol |
Luminous intensity | candela | cd |
Key Takeaways
The scientific method is the foundation of physics, emphasizing observation, hypothesis, prediction, and testing.
Symmetry is a guiding principle in formulating physical laws.
Matter, energy, space, and time are the fundamental components of the universe.
Physical quantities must be expressed with both a value and a unit.
Significant digits reflect the precision of measurements.
Unit conversions and understanding density are essential quantitative tools in physics.
