 Back
BackHeat and Thermodynamics: Structured Study Notes for PHY 103
Study Guide - Smart Notes

Temperature, Heat, and Thermodynamic Laws
Key Concepts in Heat and Thermodynamics
Heat and thermodynamics are foundational topics in physics, describing energy transfer, temperature, and the behavior of matter. Understanding these concepts is essential for analyzing physical systems and their energy exchanges.
Temperature: A measure of the average kinetic energy of particles in a substance.
Heat: Energy transferred between objects due to a temperature difference.
Thermal Contact: When two objects can exchange energy.
Thermal Equilibrium: When two objects in contact no longer exchange energy.
Zeroth Law of Thermodynamics: If two objects are each in thermal equilibrium with a third, they are in equilibrium with each other.
Temperature Scales and Thermometers
Temperature can be measured using various scales and devices, each based on physical properties that change with temperature.
Kelvin (Thermodynamic) Scale: Absolute scale; ice point at 273.15 K, triple point at 273.16 K, steam point at 373.15 K.
Celsius Scale: Ice point at 0°C, steam point at 100°C, divided into 100 intervals.
Fahrenheit Scale: Ice point at 32°F, steam point at 212°F, divided into 180 intervals.
Thermometers: Devices that measure temperature using thermometric properties (e.g., liquid expansion, gas pressure, electrical resistance).
Conversion Formulas:
Defining a Temperature Scale: Choose a substance, select a thermometric property, measure at two fixed points, and assign values using:
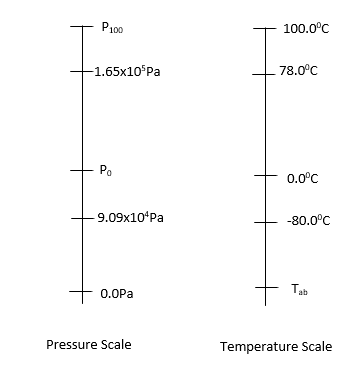
Thermal Expansion of Solids and Liquids
Thermal Expansion Principles
When temperature increases, most solids and liquids expand due to increased atomic separation. This phenomenon is crucial in engineering and construction.
Linear Expansion:
Volume Expansion:
Area Expansion:
For solids: ,
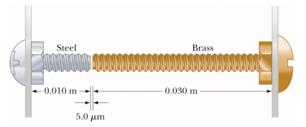
Example: Expansion joints in bridges and railways accommodate thermal expansion.
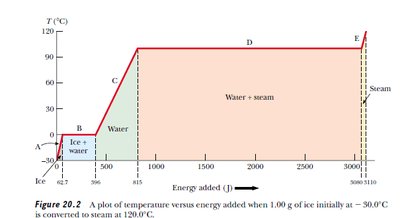
Heat Capacity, Specific Heat, and Latent Heat
Heat Capacity and Specific Heat
Heat capacity () is the energy needed to raise the temperature of a sample by 1°C. Specific heat () is the energy needed to raise 1 kg of a substance by 1°C.
Latent Heat
Latent heat () is the energy required for a phase change without temperature change.
Types: Latent heat of fusion (melting), vaporization (boiling), sublimation (solid to gas).
Ideal Gas Law and Kinetic Theory
Macroscopic Description of an Ideal Gas
An ideal gas follows the equation of state, relating pressure, volume, and temperature:
Kinetic Theory of Gases
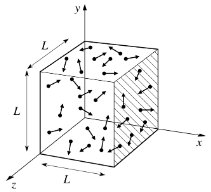
The kinetic theory explains gas pressure as a result of molecular collisions. Assumptions include large numbers of molecules, random motion, elastic collisions, and negligible intermolecular forces except during collisions.
Pressure is proportional to the number of molecules per unit volume and their average kinetic energy.
Average kinetic energy per molecule:
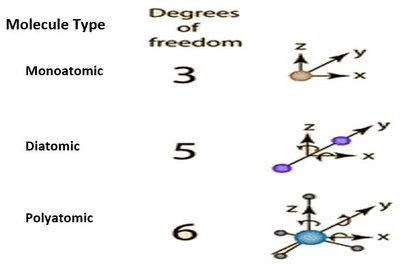
Degrees of freedom: Monoatomic (3), Diatomic (5), Polyatomic (6)
First Law of Thermodynamics
Energy Conservation in Thermodynamic Systems
The first law states that the change in internal energy () of a system is equal to the heat added () minus the work done ():
Positive : energy enters system; positive : work done by system.
Special Thermodynamic Processes
Isolated System: No heat or work exchange; internal energy constant.
Cyclic Process: Returns to initial state; internal energy change is zero.
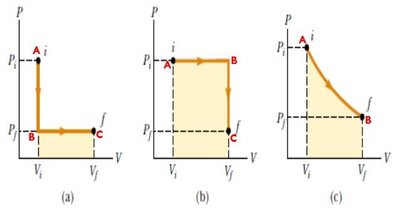
Isobaric Process: Constant pressure;
Isochoric Process: Constant volume; ,
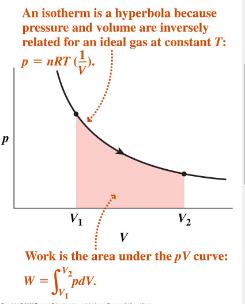
Isothermal Process: Constant temperature; ,
Second Law of Thermodynamics and Heat Engines
Heat Engines and Efficiency
A heat engine converts heat energy into work, operating between a hot and cold reservoir. Efficiency () is the ratio of work output to heat input:
The second law states that no engine can convert all heat into work; some energy is always expelled to the cold reservoir.
Heat Pumps and Refrigerators
These devices transfer heat from cold to hot reservoirs, requiring work input. Their performance is measured by the coefficient of performance (CoP):
Cooling mode:
Heating mode:
Carnot Engine and Maximum Efficiency
The Carnot engine is an ideal, reversible heat engine. Its efficiency depends only on the temperatures of the reservoirs:
Maximum efficiency is achieved only if the cold reservoir is at absolute zero, which is impossible in practice.
Third Law of Thermodynamics
Absolute Zero and Entropy
The third law states that the entropy of a perfect crystal at absolute zero is zero. Absolute zero (0 K) is unattainable experimentally, and at this temperature, all molecular motion ceases.
Entropy formula:
Energy Transfer Mechanisms
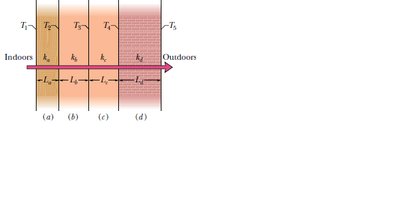
Conduction
Heat transfer by conduction occurs as molecules in a hot region transfer energy to adjacent cooler regions. The rate of energy transfer is given by:
Thermal resistance:
Convection
Convection occurs as heated fluid rises due to decreased density, transferring energy throughout the fluid.
Radiation
All objects emit energy as electromagnetic waves. The rate of energy emission is given by Stefan-Boltzmann law:
Summary Table: Temperature Scales
Scale | Ice Point | Steam Point | Interval | Conversion |
|---|---|---|---|---|
Kelvin | 273.15 K | 373.15 K | 100 | |
Celsius | 0°C | 100°C | 100 | |
Fahrenheit | 32°F | 212°F | 180 |
Summary Table: Types of Thermometers
Type | Thermometric Property | Range |
|---|---|---|
Liquid-in-glass | Length of liquid column | -35°C to 350°C (mercury) |
Gas (constant volume) | Pressure | -270°C to 1500°C |
Resistance | Electrical resistance | -200°C to 1000°C (platinum) |
Thermocouple | EMF | -270°C to 2000°C |
Summary Table: Energy Transfer Mechanisms
Mechanism | Description | Formula |
|---|---|---|
Conduction | Direct transfer via collisions | |
Convection | Bulk movement of fluid | -- |
Radiation | Electromagnetic emission |
