 Back
BackHeat, Density, and Thermodynamics: Study Notes
Study Guide - Smart Notes

Heat and Thermodynamics
Density
Density is a fundamental property of matter that describes how much mass is contained within a given volume. It is a measure of how closely packed the particles are in a substance.
Definition: Density is defined as mass divided by volume.
Formula:
Interpretation: The closer together the particles, the higher the density; the farther apart, the lower the density.
Comparison: Solids generally have higher densities than liquids, and liquids have higher densities than gases, though there are exceptions.
Application: Objects with greater density are more likely to sink, while those with lower density are more likely to float.
Standard: The density of water is 1 g/ml (or 1 g/cm3), used as a reference for other substances.
Example: Comparing two boxes of particles, the box with more closely packed particles has higher density.
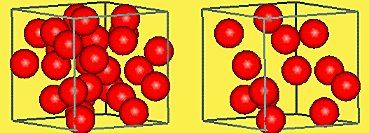
Changes in Density
Density can change with temperature. For most materials, as temperature increases, density decreases because particles move farther apart.
Exception: Water is a notable exception; ice is less dense than liquid water, which is why ice floats.
Thermal Energy
Definition and Properties
Thermal energy is the energy associated with the vibration and movement of particles within a substance. The faster the particles move, the greater their kinetic energy (KE).
Temperature: A measure of the average kinetic energy of the particles in a body.
Relationship: Faster moving particles correspond to higher temperature.
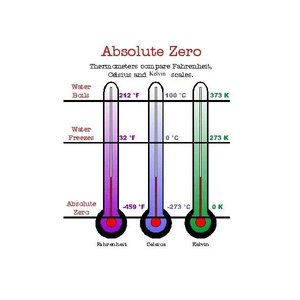
Temperature Scales
Temperature can be measured using different scales: Fahrenheit, Celsius (Centigrade), and Kelvin.
Absolute Zero: At 0 K (Kelvin), atomic motion stops, representing the lowest possible temperature.
Note: Even at absolute zero, electrons continue to orbit the nucleus.
Heat
Definition and Transfer Methods
Heat is thermal energy in motion, transferring from one place to another. There are three primary methods of heat transfer:
Conduction: Transfer of thermal energy through direct collisions between particles. Most effective in solids.
Convection: Transfer of energy through the bulk motion of materials with different densities, typically in liquids and gases.
Radiation: Transfer of energy via electromagnetic waves (light), which does not require a medium.
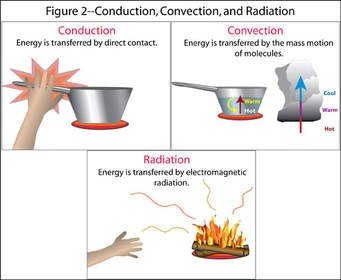
Examples of Heat Transfer
Conduction: A spoon in hot chocolate gets hot.
Convection: All of the soup in a pot gets hot.
Radiation: A heat lamp keeps a burger hot.
Laws of Thermodynamics
Overview
Thermodynamics is the study of heat and its transformation into different forms of energy. The field is governed by three fundamental laws.
First Law of Thermodynamics
The first law, also known as the conservation of energy, states that energy cannot be created or destroyed, only transformed from one form to another.
Mathematical Expression: Where is the change in internal energy, is heat added to the system, and is work done by the system.
Implication: Heat added increases thermal energy; heat removed decreases it.

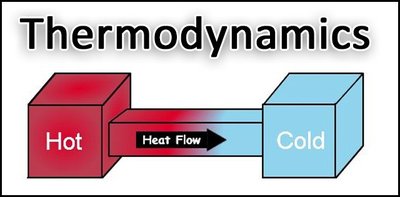
Second Law of Thermodynamics
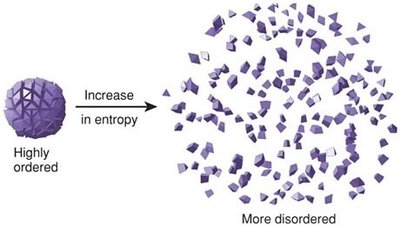
The second law states that heat naturally flows from higher temperature to lower temperature, and systems tend toward greater disorder (higher entropy).
Entropy: A measure of the randomness or disorder in a system. As energy is used, entropy increases.
Implication: Systems evolve from ordered to disordered states.
Third Law of Thermodynamics
The third law states that the entropy of a perfect crystal at absolute zero is zero, and no system can reach absolute zero.
Implication: Absolute zero is unattainable in practice.

Summary Table: Methods of Heat Transfer
Method | Medium Required | Mechanism | Example |
|---|---|---|---|
Conduction | Solid | Direct particle collisions | Spoon in hot chocolate |
Convection | Liquid/Gas | Bulk motion of material | Soup in a pot |
Radiation | None | Electromagnetic waves | Heat lamp |
Key Terms and Concepts
Density: Mass per unit volume
Thermal Energy: Energy from particle motion
Temperature: Average kinetic energy of particles
Heat: Thermal energy in transit
Conduction, Convection, Radiation: Methods of heat transfer
Entropy: Degree of disorder in a system
Thermodynamics: Study of heat and energy transformations
Additional info: Academic context was added to clarify the laws of thermodynamics, entropy, and the mathematical expression of the first law.
