 Back
BackHeat Engines and Refrigerators: Principles, Efficiency, and Thermodynamic Limits
Study Guide - Smart Notes

Chapter 21: Heat Engines and Refrigerators
Introduction to Thermodynamics
Thermodynamics is the branch of physics that studies the transformation of energy, especially the conversion between heat and work. Devices such as engines and refrigerators are practical applications of thermodynamic principles, governed by the first and second laws of thermodynamics.
First Law of Thermodynamics: Energy is conserved; the change in thermal energy of a system is equal to the work done on the system plus the heat added: .
Second Law of Thermodynamics: Most macroscopic processes are irreversible. Heat energy spontaneously transfers from a hotter system to a colder system, but never the reverse.
Heat Engines
Definition and Operation
A heat engine is a device that transforms heat energy into useful work by operating in a cyclical process. It requires two energy reservoirs: a hot reservoir (source of heat) and a cold reservoir (sink for waste heat). The engine absorbs heat from the hot reservoir, does work, and expels some heat to the cold reservoir.
Heat engines follow a cyclical process, often represented on a pV diagram or energy-transfer diagram.
They are governed by the first and second laws of thermodynamics.
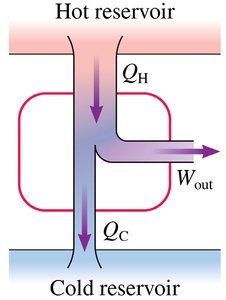
Work Done by the System
The work done by the system () is the area under the pV curve for a given process. In terms of , the first law is:

Energy Reservoirs
An energy reservoir is a large object or environment whose temperature does not change when heat is transferred. The hot reservoir is at a higher temperature than the system, while the cold reservoir is at a lower temperature.
Thermal Efficiency
The thermal efficiency () of a heat engine measures how well it converts heat into work:
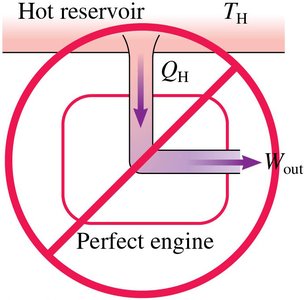
Actual engines have to (10% to 50%).
No engine can have due to energy conservation.
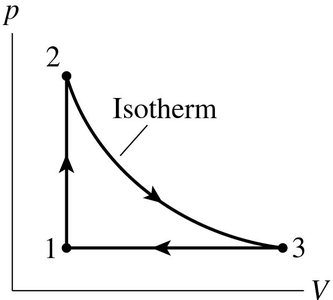
pV Diagrams and Cycles
Heat engines are often analyzed using pV diagrams, where the area enclosed by the cycle represents the net work done per cycle.
Example: Steam Turbine
In a modern power plant, steam expands to spin a turbine, then is condensed and pumped back to the boiler. This cyclical process is a practical example of a heat engine.

Refrigerators and Heat Pumps
Definition and Operation
A refrigerator (or heat pump) is a device that uses external work to transfer heat from a cold reservoir to a hot reservoir, essentially operating as a heat engine in reverse.
Requires work input () to move heat "uphill" from cold to hot.
The coefficient of performance () measures efficiency:
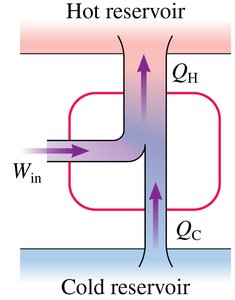
Limits of Refrigeration
No perfect refrigerator exists (i.e., is impossible).
The second law forbids spontaneous heat flow from cold to hot without work input.
Ideal-Gas Heat Engines and Processes
Ideal-Gas Cycles
Some heat engines use an ideal gas as the working substance. Their cycles can be represented as closed loops on a pV diagram. The net work done is the area inside the loop.
Summary of Ideal-Gas Processes
Process | Gas Law | Work | Heat | Thermal Energy |
|---|---|---|---|---|
Isochoric | 0 | |||
Isobaric | ||||
Isothermal | ||||
Adiabatic | See text | 0 |
Properties of Monatomic and Diatomic Gases
Monatomic | Diatomic | |
|---|---|---|
Thermodynamic Cycles: Brayton and Carnot
The Brayton Cycle
The Brayton cycle is used in jet engines and involves adiabatic compression, isobaric heating, adiabatic expansion, and isobaric cooling. Its efficiency depends on the pressure ratio:
where
The Carnot Cycle
The Carnot cycle is an idealized engine cycle that achieves the maximum possible efficiency between two temperature reservoirs. It consists of two isothermal and two adiabatic processes.
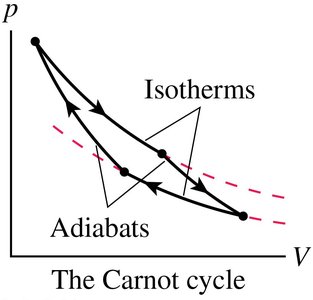
Maximum efficiency:
Maximum coefficient of performance for refrigerators:
Limits of Efficiency and the Second Law
Second Law and Carnot's Theorem
No engine operating between two reservoirs can be more efficient than a Carnot engine.
No refrigerator can have a coefficient of performance greater than a Carnot refrigerator.
Perfect engines () and perfect refrigerators () are impossible.
Applications and Problem-Solving Strategy
Steps for Analyzing Heat Engines and Refrigerators
Identify each process in the cycle.
Draw the pV diagram of the cycle.
Determine , , and at the beginning and end of each process.
Calculate , , and for each process.
Determine or , , and .
Calculate or .
Verify for a complete cycle.
Summary Table: Key Formulas
Quantity | Formula |
|---|---|
Thermal Efficiency (Heat Engine) | |
Carnot Efficiency | |
Coefficient of Performance (Refrigerator) | |
Carnot Coefficient of Performance |
Conclusion
Understanding the principles of heat engines and refrigerators is essential for analyzing and optimizing devices that convert energy between heat and work. The laws of thermodynamics set fundamental limits on efficiency, with the Carnot cycle representing the theoretical maximum. Real-world applications, such as power plants and refrigerators, operate below these limits due to practical constraints.
