 Back
BackHeat Transfer by Radiation and the Stefan-Boltzmann Law
Study Guide - Smart Notes

Heat Transfer by Radiation
Introduction to Radiation Transfer
Heat transfer by radiation is a fundamental mechanism in thermodynamics, where energy is transferred between objects via electromagnetic waves. Unlike conduction and convection, radiation does not require a medium and can occur in a vacuum. The amount of energy transferred depends on the temperature, surface area, and properties of the material.
Electromagnetic radiation is the means by which heat is transferred from one object to another.
Emissivity (ε) is a measure of how efficiently a surface emits or absorbs radiation, ranging from 0 (perfect reflector) to 1 (perfect blackbody).
A blackbody is an idealized object that absorbs and emits all incident electromagnetic radiation.
The Stefan-Boltzmann Law
The Stefan-Boltzmann law quantifies the power radiated from a surface as a function of its temperature and emissivity. This law is essential for understanding energy transfer in thermodynamic systems.
Stefan-Boltzmann Equation: The rate of energy leaving or being absorbed by an object of temperature T (in kelvin) and area A is given by:
σ (Stefan-Boltzmann constant):
ε (Emissivity):
For a perfect blackbody, .
Net Rate of Energy Transfer by Radiation
When an object is surrounded by other bodies at different temperatures, the net rate of energy transfer is the difference between the energy emitted and absorbed:
Good absorbers are also good emitters of heat.
Examples and Applications
Example 1: Electric Furnace Blackbody Radiation
An electric furnace with a small opening acts almost like a blackbody. If the opening has an area of 150 mm2 and the furnace is held at 1250°C, the power radiated is calculated using the Stefan-Boltzmann law.
Convert temperature to kelvin:
Area:
For a blackbody ():
Substitute values to find the radiated power.
Example 2: Household Radiator
A household radiator with an emissivity of 0.53 and area 1.65 m2 emits and absorbs radiation depending on its temperature and the temperature of the surroundings.
(a) Energy emitted at 53.0°C ():
(b) Energy absorbed from walls at 22.5°C ():
(c) Net rate of energy radiated:
Emissivity and Surface Properties
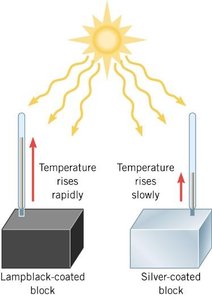
The rate at which an object absorbs or emits radiation depends strongly on its surface properties. Dark, matte surfaces (like lampblack) have high emissivity, while shiny, metallic surfaces (like silver) have low emissivity.
Lampblack-coated block: High emissivity, temperature rises rapidly.
Silver-coated block: Low emissivity, temperature rises slowly.
Thermal Imaging and Radiation
Thermal imaging is a practical application of radiation transfer, allowing visualization of temperature distributions based on emitted infrared radiation. Objects at different temperatures emit varying amounts of infrared radiation, which can be detected and displayed as color-coded images.
Thermal images show regions of higher and lower temperature, illustrating the concept of radiative heat transfer.


Important Equations Summary
Stefan-Boltzmann Law:
Net Radiation Transfer:
Additional info:
Radiative heat transfer is significant in astrophysics, climate science, and engineering applications such as furnace design and thermal insulation.
Emissivity can be engineered for specific purposes, such as minimizing heat loss in spacecraft or maximizing heat absorption in solar panels.
