 Back
BackIntroduction to Electric Charge and Electrostatics
Study Guide - Smart Notes

Structure of Matter and Fundamental Forces
Overview of the Universe's Structure
The universe is composed of matter that interacts through four fundamental forces. Understanding these forces is essential for explaining a wide range of physical phenomena, from the behavior of atoms to the motion of planets.
Gravity: The force of attraction between masses, governing planetary motion and the structure of the universe.
Electromagnetism: The force responsible for electric and magnetic interactions, including light and electricity.
Strong Force: The force that holds protons and neutrons together in the atomic nucleus.
Weak Force: Responsible for radioactive decay and certain nuclear reactions.



Earth as a Magnetized Object
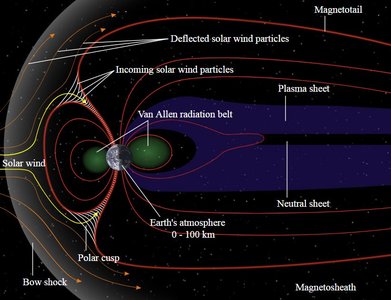
Earth's Magnetic Properties
The Earth behaves as a giant magnet due to the movement of molten iron in its outer core. This magnetic field protects the planet from solar wind and cosmic radiation, and is responsible for phenomena such as the aurora borealis.
Magnetosphere: The region around Earth dominated by its magnetic field.
Van Allen Belts: Zones of charged particles trapped by Earth's magnetic field.
Auroras: Light displays caused by charged particles interacting with the atmosphere near the poles.


Electrostatic Interactions
Nature of Electrostatic Forces
Electrostatic interactions arise from the presence and movement of electric charges. These forces are responsible for phenomena such as static electricity and lightning.
Like charges repel, unlike charges attract.
Electrostatic effects are commonly observed when materials are rubbed together, transferring electrons and creating charge imbalances.

Electrostatic Interactions and the Triboelectric Series
Charging by Friction
When two different materials are rubbed together, electrons may transfer from one to the other, resulting in one material becoming positively charged and the other negatively charged. The tendency of a material to gain or lose electrons is described by the triboelectric series.
Example: Rubbing a glass rod with silk transfers electrons from the glass to the silk, charging the glass positively and the silk negatively.
Material | Charge Tendency |
|---|---|
Glass | Positive |
Silk | Negative |
Rubber | Negative |
Wool | Positive |

Electric Charge
Properties of Electric Charge
Electric charge is a fundamental property of matter, carried by elementary particles such as electrons and protons. It is quantized and conserved in all physical processes.
Positive and Negative Charges: Like charges repel, unlike charges attract.
Electrical Neutrality: Objects with equal numbers of positive and negative charges are neutral.
Conservation of Charge: The total charge in a closed system remains constant.

Quantization of Charge
Electric charge exists in discrete units, with the elementary charge being the charge of a single proton or electron.
Quantization Formula:
where
Elementary charge: C
Coulomb (C): The SI unit of charge. One coulomb is the amount of charge transferred by a current of one ampere in one second.

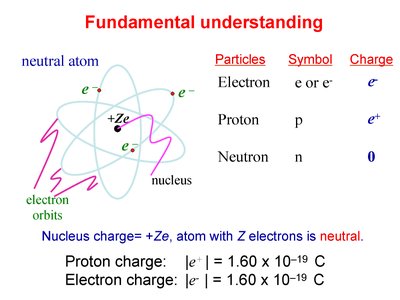
Atomic Structure and Charge
Fundamental Particles and Their Charges
Atoms are composed of protons, neutrons, and electrons. The nucleus contains protons and neutrons, while electrons orbit the nucleus. The overall charge of an atom is determined by the balance of protons and electrons.
Particle | Symbol | Charge |
|---|---|---|
Electron | e or e- | -e |
Proton | p | +e |
Neutron | n | 0 |
Proton charge: C
Electron charge: C
Conductors, Insulators, and Related Materials
Classification of Materials by Electrical Properties
Materials are classified based on their ability to allow electric charges to move freely.
Conductors: Allow free movement of charges (e.g., metals, tap water, the human body).
Insulators (Dielectrics): Do not allow free movement of charges (e.g., air, glass, plastic).
Semi-conductors: Intermediate properties; conduct under certain conditions (e.g., silicon, germanium).
Super-conductors: Exhibit zero resistance to charge movement under specific conditions.

Behavior of Conductors and Insulators Near Charges
When a charged object is brought near a conductor, free electrons redistribute to balance the charge. In insulators, charges remain fixed in place.
Example: Bringing a negatively charged rod near a metal ball causes electrons in the ball to move away from the rod, leaving one side positively charged.

Electroscope
Measuring Electric Charge
An electroscope is a device used to detect the presence and magnitude of electric charge. The angle of deflection of its needle or leaves is proportional to the amount of charge present.
Operation: When a charged object touches or comes near the electroscope, free electrons move, causing the needle to deflect.

Polarization of Insulators
Induced Charge Separation
In insulators, atoms can become polarized when exposed to an external electric field, even though free electrons are not present. This polarization can lead to weak attractive forces between the insulator and a charged object.
Example: Bringing a charged rod near a plastic or glass object causes the atoms to align, creating a polarized region.

Polarization of Conductors and Insulators
Comparison of Charge Behavior
Conductors and insulators respond differently to external electric fields. In conductors, charges separate and move freely, while in insulators, atoms become polarized but do not exhibit net charge separation.
Example: Metal cups become oppositely charged when separated after polarization; plastic cups do not retain charge separation.

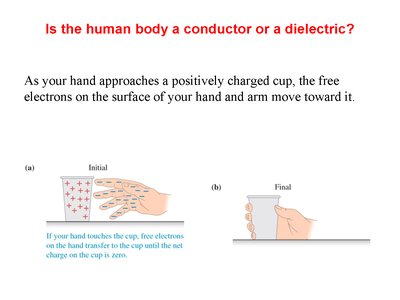
The Human Body: Conductor or Dielectric?
Electrical Properties of the Human Body
The human body acts as a conductor because it contains free ions and water, allowing electrons to move freely. When a hand approaches a charged object, electrons redistribute accordingly.
Example: Touching a positively charged cup allows electrons from the hand to neutralize the cup's charge.
Grounding
Neutralizing Electric Charge
Grounding provides a path for excess charge to flow between an object and the Earth, neutralizing the object's charge. This is a common safety measure in electrical systems.
Example: Connecting a positively charged cup to the ground allows electrons to flow from the Earth to the cup, making it neutral.

