 Back
Backlecture 26
Study Guide - Smart Notes

Nuclear Physics and Radioactivity
Radioactive Decay and Activity
Radioactive decay is a spontaneous process in which unstable atomic nuclei lose energy by emitting radiation. The rate at which a radioactive substance decays is characterized by its half-life, which is the time required for half of the radioactive nuclei in a sample to disintegrate.
Half-life (T1/2): The time in which half of the original number of radioactive nuclei decay.
Decay Law: The number of undecayed nuclei at time t is given by: where is the initial number of nuclei and is the decay constant.
Relationship between half-life and decay constant:
Activity (A): The rate at which nuclei decay, measured in becquerels (Bq), where 1 Bq = 1 decay/s.
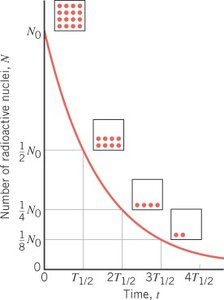
Example: If a sample starts with nuclei, after one half-life, ; after two half-lives, , and so on.
Units of Activity
Becquerel (Bq): SI unit, 1 Bq = 1 decay/s
Curie (Ci): 1 Ci = Bq
Radioactive Dating
Radioactive dating uses the known half-life of radioactive isotopes to determine the age of objects. Carbon-14 dating is commonly used for organic remains.
Key Equation: The age t of a sample can be found using:
Application: Used to date archaeological finds, such as the "Ice Man" discovered in the Alps.
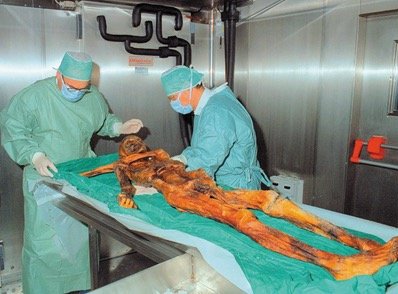
Example: If the activity per gram of carbon in a living organism is 0.23 Bq and in a sample is 0.121 Bq, the age can be calculated as shown above.
Radioactive Decay Series
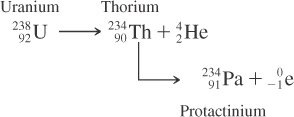
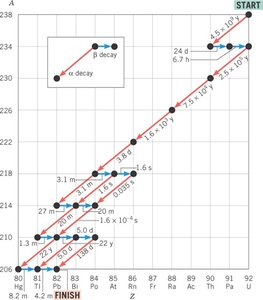
Some heavy nuclei decay through a series of steps, producing a sequence of different elements until a stable nucleus is formed. This is known as a radioactive decay series.
Decay Series: Sequential decay of one nucleus after another, often involving both alpha and beta decays.
Example: The uranium-238 decay series eventually leads to stable lead-206.
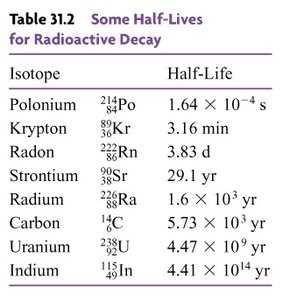
Half-Lives of Common Isotopes
The half-life of a radioactive isotope is a crucial property that determines its usefulness in dating and other applications.
Isotope | Half-Life |
|---|---|
Polonium-214 | s |
Krypton-89 | 3.16 min |
Radon-222 | 3.83 d |
Strontium-90 | 29.1 yr |
Radium-226 | yr |
Carbon-14 | yr |
Uranium-238 | yr |
Indium-115 | yr |
Nuclear Energy, Effects, and Uses of Radiation
Induced Nuclear Reactions
Nuclear reactions occur when a nucleus is struck by another nucleus, particle, or photon, causing a change in the target nucleus. Both charge and nucleon number are conserved in these reactions.
General Form: or
Q-value: The energy released or absorbed in a reaction:
Conservation Laws: Both electric charge and nucleon number are conserved.
Nuclear Reaction | Notation |
|---|---|

Conserved Quantity | Before Reaction | After Reaction |
|---|---|---|
Total electric charge (number of protons) | 2 + 13 | Z + 0 |
Total number of nucleons | 4 + 27 | A + 1 |

Nuclear Fission
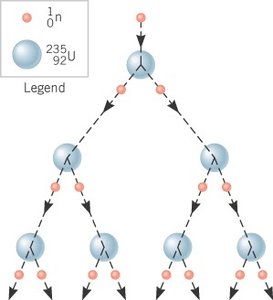
Nuclear fission is the splitting of a heavy nucleus into two lighter nuclei, accompanied by the release of energy and additional neutrons. This process can lead to a chain reaction.
Example: fission products (e.g., barium, krypton) + 3 neutrons + energy (~200 MeV)
Chain Reaction: The released neutrons can induce further fissions, leading to a self-sustaining chain reaction.
Controlled vs. Uncontrolled: In reactors, the chain reaction is controlled; in atomic bombs, it is uncontrolled.
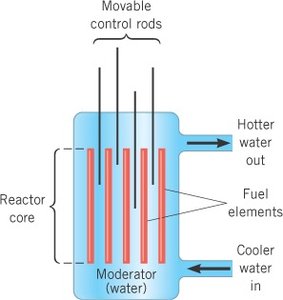
Nuclear Reactors
Nuclear reactors use controlled fission chain reactions to generate energy. Key components include fuel elements, control rods, and a moderator.
Fuel: Usually enriched uranium-235.
Moderator: Slows down neutrons to increase the probability of fission (often water).
Control Rods: Absorb neutrons to regulate the chain reaction (often made of boron).
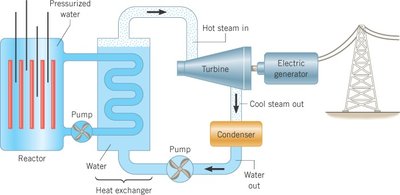
The efficiency of converting nuclear energy to electricity is about 1/3, with energy losses in heat exchange and mechanical processes.
Nuclear Fusion
Nuclear fusion is the process by which two light nuclei combine to form a heavier nucleus, releasing energy. Fusion powers stars, including the Sun.
Example Reaction: Energy released: MeV
Proton-Proton Chain: The main fusion process in the Sun, converting hydrogen to helium and releasing energy.
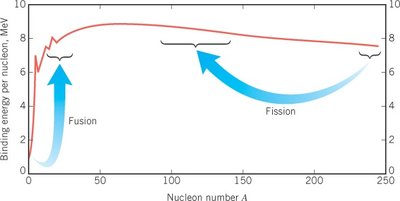
Fusion vs. Fission: Fusion combines light nuclei (high binding energy per nucleon), while fission splits heavy nuclei (lower binding energy per nucleon), both releasing energy due to the difference in binding energy.
Additional info: These notes cover the fundamental concepts of nuclear physics and radioactivity, including decay laws, dating methods, nuclear reactions, fission, fusion, and their applications in energy production.
