 Back
BackNuclear Physics: Structure, Stability, Binding Energy, and Radioactivity
Study Guide - Smart Notes

Nuclear Physics
Atomic Nuclei: Structure and Constituents
The atomic nucleus is the dense central core of an atom, composed of protons and neutrons (collectively called nucleons). Protons carry a positive charge, while neutrons are electrically neutral. Electrons, which are negatively charged, orbit the nucleus at much greater distances, making the atom mostly empty space. Nearly all the mass of an atom is concentrated in its nucleus, which has a typical size on the order of m (or nm).
Proton number (Z): The number of protons in the nucleus, which determines the chemical element.
Neutron number (N): The number of neutrons in the nucleus.
Mass number (A): The total number of nucleons, .
Example: Carbon-12 (C) has 6 protons and 6 neutrons; Uranium-238 (U) has 92 protons and 146 neutrons.
Isotopes
Isotopes are atoms of the same element (same Z) but with different numbers of neutrons (N), resulting in different mass numbers (A). Isotopes of an element have nearly identical chemical properties but may have different nuclear properties, such as stability.
Isotope notation: Element (e.g., C, Pb, U)
Example: U (Uranium-238) has 92 protons and 146 neutrons ().
Atomic Mass and Mass Unit
The atomic mass unit (u) is a standard unit for expressing atomic and nuclear masses:
Energy equivalent: (using )
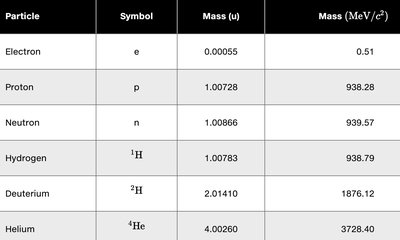
The atomic mass listed on the periodic table is the weighted average of the masses of all naturally occurring isotopes of that element.
Nuclear Size and Density
Nuclei are extremely dense and have a sharp boundary. The radius of a nucleus depends on its mass number (A):
, where
Nuclear density is approximately , much greater than ordinary matter.
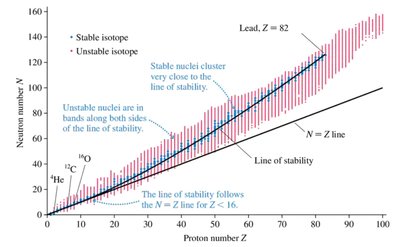
Nuclear Stability
Most isotopes are radioactive and unstable, but about 252 isotopes are stable. Stability depends on the ratio of neutrons to protons (N/Z). For light elements, stability occurs near , but for heavier elements, more neutrons are needed for stability. No nuclei with (lead) are stable.
Nuclear Binding Energy
The nuclear binding energy is the energy required to separate all nucleons in a nucleus. It is calculated from the mass defect, which is the difference between the mass of the nucleus and the sum of the masses of its individual protons and neutrons:
Binding energy per nucleon:
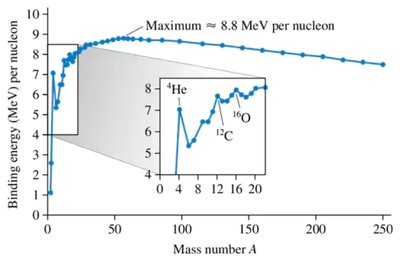
Binding energy per nucleon peaks at about 8 MeV for medium-mass nuclei (A ≈ 60), indicating maximum stability. Heavier nuclei can become more stable by fission (splitting), and lighter nuclei by fusion (combining).
The Strong Nuclear Force
The strong force is the fundamental force that holds nucleons together in the nucleus, overcoming the electrostatic repulsion between protons. It is:
Attractive between all nucleons (proton-proton, neutron-neutron, proton-neutron)
Short-ranged (acts over distances ~1-3 fm)
Much stronger than the electromagnetic force at nuclear distances
Neutrons help stabilize the nucleus by providing additional strong force attraction without adding to the electrostatic repulsion.
Radioactivity and Nuclear Decay
Radioactivity is the spontaneous emission of particles or electromagnetic radiation from unstable nuclei. The number of radioactive nuclei decays exponentially with time:
, where is the mean lifetime
The half-life is the time for half the nuclei to decay:
Alternative form:
Nuclear Activity
The activity (R) of a radioactive sample is the number of decays per second:
SI unit: becquerel (Bq), where 1 Bq = 1 decay/second
Older unit: curie (Ci), where 1 Ci = Bq
Radioactive Dating
Radioactive isotopes are used to date materials. For example, C (carbon-14) dating is used for organic materials, with a half-life of 5730 years. The ratio of C to C in a sample can be used to estimate its age. Other isotopes, such as K, are used for dating rocks.
Nuclear Decay Mechanisms
Alpha decay: Emission of an alpha particle (He nucleus), reducing A by 4 and Z by 2.
General form:
Beta decay: Emission of an electron (beta minus) or positron (beta plus), changing a neutron to a proton or vice versa.
Beta minus: ,
Beta plus: ,
Gamma decay: Emission of a high-energy photon (gamma ray) as the nucleus transitions from an excited state to a lower energy state. No change in A or Z.
Decay Series
Often, the daughter nucleus from a radioactive decay is itself unstable, leading to a sequence of decays known as a decay series, until a stable nucleus is formed.
Types of Radiation
There are three main types of nuclear radiation:
Radiation | Identification | Charge | Stopped by |
|---|---|---|---|
Alpha, | He nucleus | +2e | Sheet of paper |
Beta, | Electron | −e | Few mm of aluminum |
Gamma, | High-energy photon | 0 | Many cm of lead |
Ionizing Radiation and Radiation Dose
Ionizing radiation has enough energy to remove electrons from atoms or break molecular bonds, causing damage to biological tissue. The absorbed dose is the energy deposited per unit mass (SI unit: gray, Gy; 1 Gy = 1 J/kg). The dose equivalent accounts for the biological effectiveness of different types of radiation and is measured in sieverts (Sv):
Dose equivalent: (absorbed dose) × (relative biological effectiveness, RBE)
1 Sv = 100 rem (old unit)
Ionizing radiation can cause mutations, cancer, or cell death, depending on the dose and exposure.
Medical Uses of Radiation
Radiation is used in medicine for both diagnosis and treatment:
Radiation therapy: Uses gamma rays or radioactive sources to kill cancer cells.
Tracers: Radioactive isotopes are used to track biological processes in the body.
Magnetic Resonance Imaging (MRI): Uses magnetic fields and radio waves to image soft tissues (not ionizing radiation).
