 Back
BackOne-Dimensional Quantum Mechanics: Study Notes
Study Guide - Smart Notes

One-Dimensional Quantum Mechanics
Introduction to Quantum Mechanics
Quantum mechanics describes the behavior of particles at atomic and subatomic scales, where classical physics fails to provide accurate predictions. The fundamental equation governing quantum systems is the Schrödinger equation, which determines the allowed energy states and the probability distributions of particles.
The Schrödinger Equation
Formulation and Interpretation
Schrödinger Equation: The time-independent Schrödinger equation for a particle of mass m in a potential U(x) is:
Wave Function (\(\psi(x)\)): Describes the quantum state of a particle. The square of its magnitude, \(|\psi(x)|^2\), gives the probability density of finding the particle at position x.
Boundary Conditions: The wave function must be continuous, normalizable, and vanish at infinity or in forbidden regions.
Quantization: Only specific energy values (eigenvalues) allow for physically meaningful solutions, leading to quantized energy levels.
de Broglie Wavelength
Particles exhibit wave-like properties, with wavelength given by:
As kinetic energy decreases, the de Broglie wavelength increases.

Solving the Schrödinger Equation
The equation is a second-order differential equation with two independent solutions. The general solution is a linear combination of these, with constants determined by boundary conditions.
Normalization ensures the total probability is one:
Problem-Solving Strategy in Quantum Mechanics
Model the system with an appropriate potential-energy function \(U(x)\).
Visualize the potential-energy curve and establish boundary conditions.
Solve the Schrödinger equation, normalize the wave function, and determine allowed energy levels.
Calculate probabilities and other physical quantities.


Particle in a Rigid Box (Infinite Potential Well)
Physical Model and Boundary Conditions
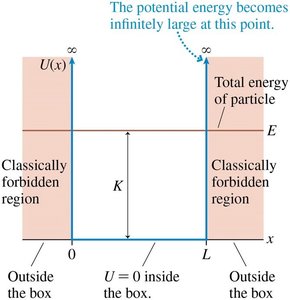
A particle of mass m is confined to a box of length L with infinitely high walls at x = 0 and x = L.
The potential energy is zero inside the box and infinite outside:
Wave Functions and Quantized Energies
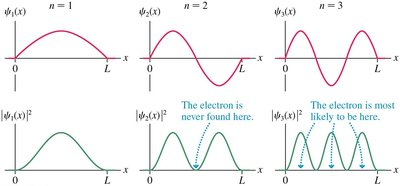
The allowed wave functions are:
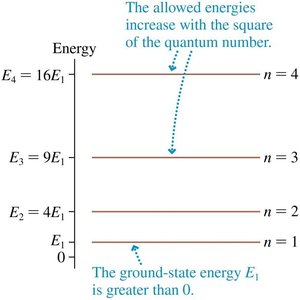
The corresponding energy levels are:
Zero-Point Energy
The lowest energy state (n = 1) is not zero; the particle cannot be at rest due to the uncertainty principle.
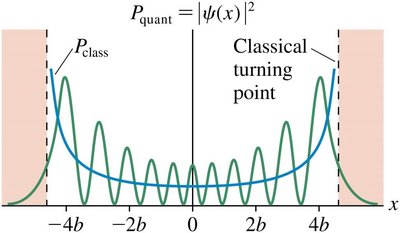
The Correspondence Principle
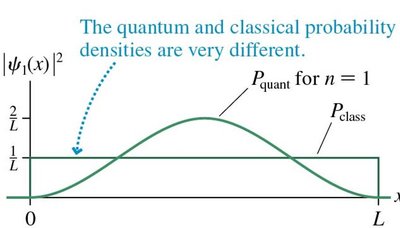
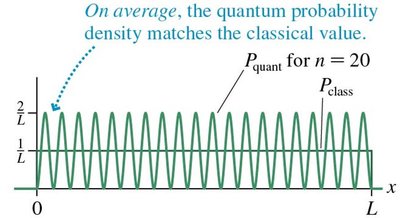
As the quantum number n increases, quantum results approach classical predictions.
For large n, the probability density \(|\psi(x)|^2\) matches the classical probability density.
Finite Potential Wells
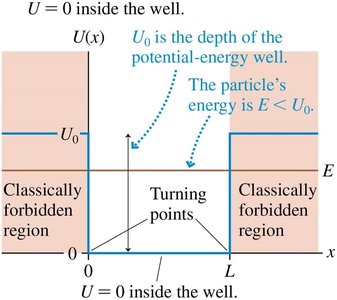
Physical Model
A particle is confined in a well of finite depth U0 and width L.
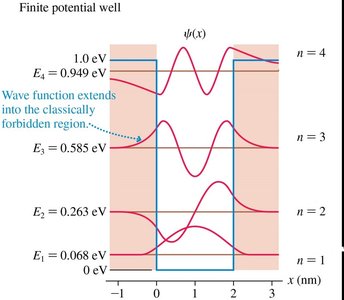
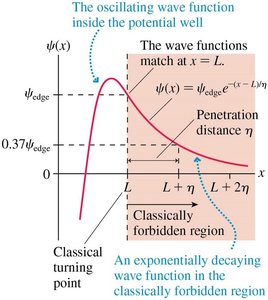
There are only a finite number of bound states, and the wave function extends into classically forbidden regions (tunneling).
Penetration Distance and Tunneling
In the forbidden region, the wave function decays exponentially:
The penetration distance \(\eta\) is:
Quantum-Mechanical Tunneling
A particle can pass through a potential barrier even if its energy is less than the barrier height—a phenomenon with no classical analog.
The probability of tunneling through a barrier of width w is:
Applications of Quantum Mechanics
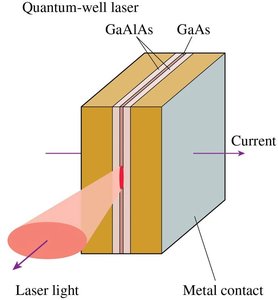
Quantum-Well Lasers
Semiconductor devices where electrons are confined in thin layers, emitting laser light when excited.

Scanning Tunneling Microscope (STM)
Uses quantum tunneling of electrons between a sharp tip and a surface to image surfaces at the atomic scale.
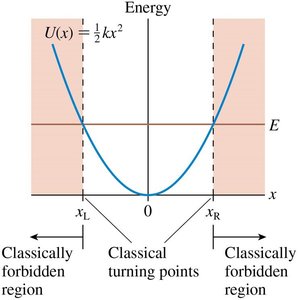
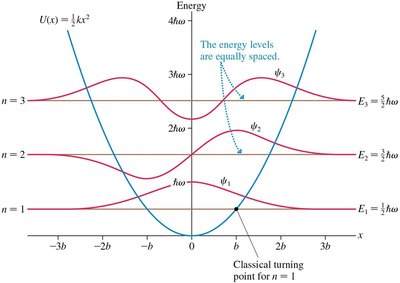
The Quantum Harmonic Oscillator
The potential energy is quadratic: .
Allowed energies are equally spaced:
Wave functions are more complex but share features with the particle in a box.
Summary Table: Key Quantum Systems
System | Potential Energy Function | Allowed Energies | Wave Function Features |
|---|---|---|---|
Particle in a Box | (inside), (outside) | Sinusoidal, zero at boundaries | |
Finite Well | (inside), (outside) | Finite number of quantized levels | Oscillatory inside, exponential decay outside |
Harmonic Oscillator | Hermite polynomials, Gaussian envelope |
Important Concepts
Wave-Particle Duality: Particles exhibit both wave-like and particle-like properties.
Quantization: Only certain energy levels are allowed in bound systems.
Tunneling: Particles can penetrate and cross potential barriers even when classically forbidden.
Correspondence Principle: Quantum results approach classical predictions for large quantum numbers.
