 Back
BackStates of Matter and Phase Transitions: Foundations of Atomic and Molecular Physics
Study Guide - Smart Notes

The Atomic Nature of Matter
Atoms: The Building Blocks of Matter
All matter is composed of atoms, which are the smallest units retaining the properties of an element. Atoms are in constant motion, attracting each other at short distances and repelling when forced together. This concept is foundational to understanding the behavior of matter in different states.

Atoms consist of a nucleus (protons and neutrons) surrounded by electrons.
Richard Feynman emphasized the centrality of atoms in all physical phenomena.
The Atomic Hypothesis: Historical Development
The idea that matter is composed of discrete units (atoms) evolved over centuries:
Ancient Greeks (5th century BC): Proposed matter is made of indivisible atoms.
Aristotle: Suggested matter was made of four elements—earth, air, fire, and water.
John Dalton (1800s): Revived atomic theory, proposing that each element consists of identical atoms.
Robert Brown (1827): Observed Brownian motion, later explained by Einstein (1905) as evidence for atoms.

Antimatter
Antimatter consists of particles with the same mass as their matter counterparts but opposite charge. For example, the antiparticle of the electron is the positron.
When matter and antimatter meet, they annihilate, releasing energy.
Antimatter cannot exist in large quantities in our universe due to this annihilation process.
Dark Matter and Dark Energy
Most of the universe is not made of ordinary matter:
Dark Matter: Interacts only gravitationally, making up about 23% of the universe's mass.
Dark Energy: A mysterious form of energy causing the accelerated expansion of the universe, comprising about 73% of its energy.
States of Matter
Classification of States
Matter exists in several states, primarily defined by the arrangement and energy of their constituent particles. The four naturally occurring states are:
Solid
Liquid
Gas
Plasma
Additional states, such as Bose-Einstein Condensates, occur under extreme conditions.
Properties of the Main States of Matter
Phase | Definite Shape? | Definite Volume? |
|---|---|---|
Solid | YES | YES |
Liquid | NO | YES |
Gas | NO | NO |
Plasma | NO | NO |
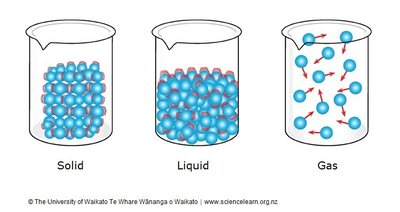
Solids
In solids, particles are tightly packed and vibrate in place. Strong intermolecular forces keep them in fixed positions.
Definite shape and volume
Particles vibrate but do not move past each other

Liquids
Liquids have particles that are close together but can slide past one another, allowing liquids to flow and take the shape of their container.
Definite volume but no definite shape
Particles are less tightly bound than in solids

Gases
In gases, particles are far apart and move freely. There are negligible forces between them, allowing gases to expand to fill any container.
No definite shape or volume
Particles move rapidly and independently

Plasma
Plasma is a state where gas particles are ionized, meaning they have lost or gained electrons and can conduct electricity. Plasmas are common in stars and certain earthly phenomena.
Examples: Lightning, neon signs, flames


Bose-Einstein Condensate (BEC)
A Bose-Einstein Condensate is formed at extremely low temperatures, where particles occupy the same quantum state and behave as a single quantum entity.
Predicted by Satyendra Nath Bose and Albert Einstein in the 1920s
First produced in 1995 using rubidium atoms cooled to near absolute zero

Phase Transitions
Energy and Phase Changes
Transitions between states of matter occur when energy is added or removed. The direction of energy flow determines the type of phase change:
Melting: Solid to liquid (energy added)
Vaporization: Liquid to gas (energy added; includes boiling and evaporation)
Ionization: Gas to plasma (energy added)
Freezing: Liquid to solid (energy removed)
Condensation: Gas to liquid (energy removed)
Deionization: Plasma to gas (energy removed)
Sublimation: Solid to gas (energy added, skipping liquid phase)
Examples of Phase Changes
Melting (Solid to Liquid): Chocolate bar melting when heated.


Vaporization (Liquid to Gas): Water boiling and turning into steam.


Condensation (Gas to Liquid): Water vapor forming raindrops.


Freezing (Liquid to Solid): Water freezing into ice or snowflakes.


Summary Table: Particle Motion and Properties by Phase
Phase | Motion of Particles | Speed of Particles |
|---|---|---|
Solid | Particles are tightly bound | Slow |
Liquid | Particles are loosely bound, can slide past one another | Medium |
Gas | Particles are independent | Fast |
Plasma | Particles are independent and ionized | Very fast |
Summary Table: Shape and Volume by Phase
Phase | Definite Shape? | Definite Volume? |
|---|---|---|
Solid | YES | YES |
Liquid | NO | YES |
Gas | NO | NO |
Plasma | NO | NO |
Key Equations and Concepts
Kinetic Molecular Theory: The temperature of a substance is proportional to the average kinetic energy of its particles.
Phase Change Energy: The energy required for a phase change is related to the breaking or forming of intermolecular forces, not to a temperature change during the transition.
Where is the heat energy, is the mass, and is the latent heat of the phase transition.
Additional info: The study of states of matter and phase transitions is foundational for understanding thermodynamics, statistical mechanics, and material science in physics.
