 Back
BackTemperature and Thermal Equilibrium: Foundations and Measurement
Study Guide - Smart Notes

Temperature and Thermal Equilibrium
Basic Definitions in Thermal Physics
Thermal physics explores the properties and behavior of systems in relation to heat and temperature. Understanding the concepts of system, surroundings, and boundary is fundamental:
System: The part of the universe under study.
Surroundings: Everything outside the system.
Boundary: The interface separating the system from its surroundings, which can be physical or imaginary.
Open system: Exchanges matter with surroundings.
Closed system: Does not exchange matter.
Diathermal boundary: Allows heat transfer.
Adiabatic boundary: Does not allow heat transfer (isolated).
Thermal Equilibrium
Thermal equilibrium is a state in which all parts of a system have uniform bulk physical properties (such as pressure, temperature, viscosity) and these properties do not change with time. When two systems are in thermal contact, heat flows from the hotter to the colder system until both reach the same temperature and heat flow ceases.
Thermal equilibrium: No net heat flow between systems; properties are uniform and stable.
Example: If a hot object is placed in contact with a cold object, heat will flow until both reach the same temperature.
The Zeroth Law of Thermodynamics
Statement and Experimental Illustration
The Zeroth Law of Thermodynamics establishes the concept of temperature and thermal equilibrium. It states: If two systems are each in thermal equilibrium with a third system, then they are in thermal equilibrium with each other. This law is foundational for defining temperature as a measurable property.
Key Point: The Zeroth Law allows the use of thermometers to compare temperatures.
Example: If system A and system B are each in equilibrium with system C, then A and B are in equilibrium with each other.
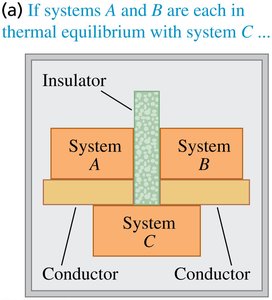
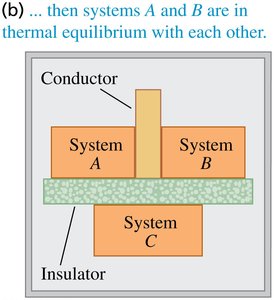
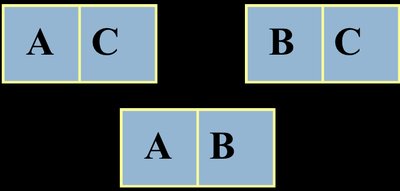
Consequences for Temperature
The Zeroth Law leads to a formal definition of temperature: Systems in thermal equilibrium with one another have the same temperature. In the microscopic view, temperature is related to the average kinetic energy of the particles in a system.
Temperature: A bulk property determining whether systems are in thermal equilibrium.
Microscopic interpretation: Higher temperature corresponds to higher average kinetic energy of particles.
Measurement of Temperature
Thermometers and Physical Properties
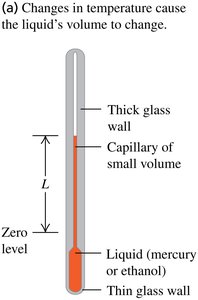
Thermometers measure temperature by exploiting physical properties that change with temperature, such as length, volume, resistance, and others. The thermometer must reach thermal equilibrium with the object being measured before an accurate reading can be taken.
Key properties used:
Length (e.g., bimetallic strip)
Volume (e.g., liquid-in-glass thermometer)
Resistance (e.g., platinum resistance thermometer)
Example: The volume of liquid in a thermometer expands with temperature.
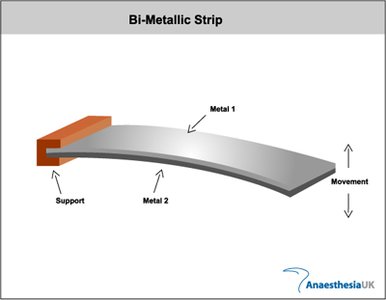
Bimetallic Strip
A bimetallic strip consists of two metals with different coefficients of thermal expansion bonded together. As temperature changes, the strip bends due to the differing expansion rates, providing a mechanical indication of temperature.
Application: Used in thermostats and temperature sensors.
Temperature Scales
Celsius, Fahrenheit, and Kelvin Scales
Temperature scales are based on reference points such as the freezing and boiling points of water. The three main scales are:
Celsius (°C): 0°C is the freezing point, 100°C is the boiling point of water.
Fahrenheit (°F): 32°F is the freezing point, 212°F is the boiling point of water.
Kelvin (K): 0 K is absolute zero, the lowest possible temperature.
Conversion formulas:
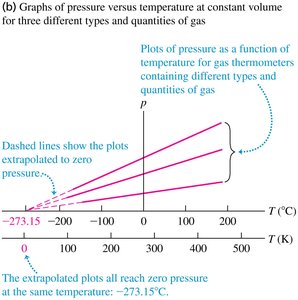
Gas Thermometers and Absolute Zero
Gas thermometers use the ideal gas law to define temperature. At constant volume, pressure is proportional to temperature:
At constant V:
Extrapolating pressure vs. temperature plots for different gases shows that all reach zero pressure at −273.15°C, defining absolute zero.
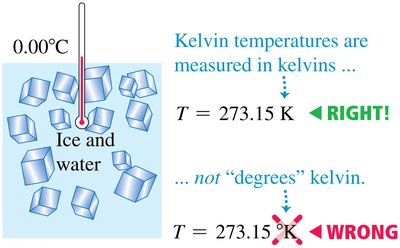
Kelvin Scale and Temperature Conversions
The Kelvin scale is the absolute temperature scale, with 0 K as absolute zero. Kelvin temperatures are not referred to as "degrees"; instead, they are simply "kelvins." The relationship between Celsius and Kelvin is:
Comparison of Temperature Scales
The following table compares key reference points across the Kelvin, Celsius, and Fahrenheit scales:
K | C | F |
|---|---|---|
Water boils: 373 | 100 | 212 |
Water freezes: 273 | 0 | 32 |
CO2 solidifies: 195 | -78 | -109 |
Oxygen liquefies: 90 | -183 | -298 |
Absolute zero: 0 | -273 | -460 |

Practical Thermometers
Types and Applications
Different types of thermometers are used depending on the temperature range and required accuracy:
Liquid expansion: Mercury, ethanol, pentane
Resistance: Platinum resistance thermometer
Thermocouple: Copper-constantan
Semiconductor: Crystalline semiconductor (especially at low T)
Carbon resistance: Useful but unreliable below 4 K
Magnetic susceptibility: Paramagnetic salts (especially below 1 K)
Pyrometer: Used at high temperatures (e.g., above 1000 K)
Each thermometer type exploits a different physical property that changes with temperature, making them suitable for specific applications and temperature ranges.
