 Back
BackThe Failure of Classical Physics and the Birth of Modern Atomic Theory
Study Guide - Smart Notes

Classical Physics at the End of the 19th Century
Overview of Classical Physics
By the late 1800s, classical physics provided a comprehensive framework for understanding the natural world, including Newton's laws of motion, Maxwell's equations for electromagnetism, and the laws of thermodynamics. However, several experimental observations could not be explained by these theories, leading to the development of modern physics.
Newton’s Laws of Motion: Describes the motion of particles under forces such as gravity, electric, and magnetic forces ().
Mechanical Waves: Includes sound and water waves.
Hamiltonian and Lagrangian Dynamics: Advanced formulations of mechanics.
Navier-Stokes Equations: Governs fluid dynamics.
Thermodynamics: Concepts of heat, entropy, and work; efficiency of engines.
Maxwell’s Equations: Unified theory of electricity and magnetism, showing that light is an electromagnetic wave.
Dalton’s Atomic Theory: Atoms as indivisible units of elements (later shown to be incorrect).
Despite its successes, classical physics could not explain certain phenomena, such as blackbody radiation and atomic spectra.
Blackbody Radiation
Properties of Blackbodies
A blackbody is an idealized object that absorbs all incident electromagnetic radiation and re-emits energy with a characteristic spectrum that depends only on its temperature. The color of a heated object (such as metal in a forge) changes with temperature, illustrating the relationship between temperature and emitted radiation.
Absorption: Black objects absorb most incident light and heat up quickly.
Emission: Hot black objects radiate energy efficiently.
Color-Temperature Relationship: The color of a blackbody is directly related to its temperature.


Stefan-Boltzmann Law
The total power radiated by a blackbody is given by the Stefan-Boltzmann law:
= emissivity (ranges from 0 to 1 for real objects)
= Stefan-Boltzmann constant ()
= surface area
= absolute temperature (in Kelvin)
Doubling the temperature increases the radiated power by a factor of 16 ().
Wien’s Displacement Law
Wien’s law relates the temperature of a blackbody to the wavelength at which it emits radiation most intensely:
As temperature increases, the peak wavelength shifts to shorter (bluer) wavelengths.
Blackbody Spectrum and the Ultraviolet Catastrophe
Classical physics (Rayleigh-Jeans law) predicted that the energy radiated at short wavelengths (ultraviolet) would become infinite—a result known as the ultraviolet catastrophe. However, experiments showed that the actual spectrum peaks and then falls off at short wavelengths.
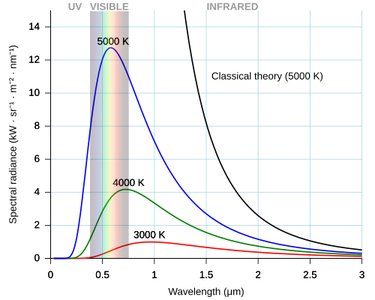
Planck resolved this by proposing that energy is quantized, leading to the birth of quantum mechanics:
Main Point: Classical physics cannot explain the shape of the blackbody emission spectrum, especially in the ultraviolet regime.
Atomic Spectra and the Failure of Classical Physics
Absorption and Emission Lines
When light passes through a gas or is emitted by excited atoms, only specific wavelengths are absorbed or emitted, producing discrete spectral lines. Each element has a unique spectral fingerprint.
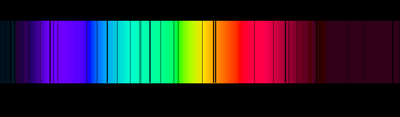
Emission Spectrum: Produced when atoms emit light at characteristic wavelengths.
Absorption Spectrum: Produced when atoms absorb light at specific wavelengths.
Classical physics could not explain why atoms emit or absorb only certain wavelengths.
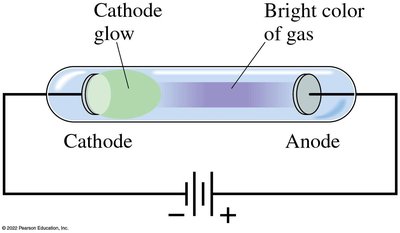
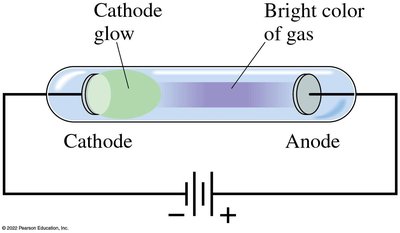
Gas Discharge Tubes and Spectral Fingerprints
Passing an electric current through a gas causes it to glow, and the color depends on the type of gas. Each atom produces a unique set of spectral lines.

Balmer Formula for Hydrogen
The Balmer formula (1885) empirically described the visible spectral lines of hydrogen:
However, the physical reason for discrete spectra was not understood until quantum mechanics.
Cathode Rays, X-Rays, and the Discovery of the Electron
Cathode Ray Tubes
When a high voltage is applied across a partially evacuated tube, a glow appears near the cathode. In low-pressure tubes, rays (cathode rays) travel from the cathode to the anode and can be deflected by magnetic and electric fields, indicating they are negatively charged particles (later called electrons).

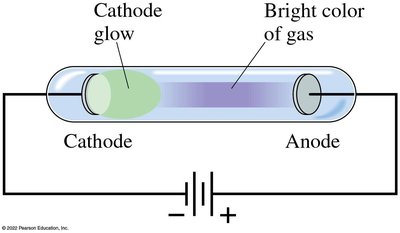
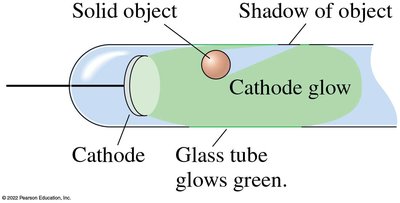
Properties of Cathode Rays
Deflected by magnetic fields (indicating charge).
Travel in straight lines and cast shadows.
Can be produced from any metal cathode.

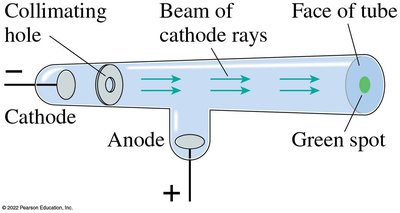
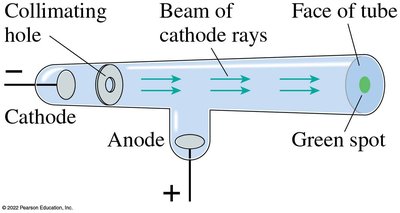
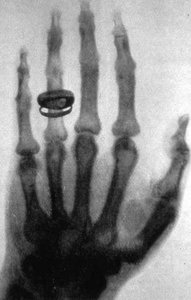
Discovery of X-Rays
Wilhelm Röntgen discovered X-rays in 1895 when he noticed that Crookes tubes emitted a new type of penetrating radiation that could pass through solids and was not deflected by magnetic fields.
Thomson’s Crossed-Field Experiment
J. J. Thomson measured the charge-to-mass ratio () of cathode rays using electric and magnetic fields. He found that $q/m$ was the same for all cathode materials and much larger than for hydrogen ions, indicating that cathode rays were much lighter than atoms and were a universal constituent of matter—electrons.
Key equations:
When the electric and magnetic forces balance, , and the charge-to-mass ratio can be determined.
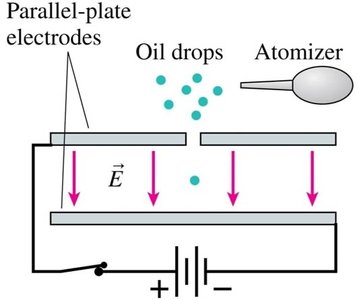
Millikan’s Oil Drop Experiment
Millikan measured the elementary charge by balancing the gravitational and electric forces on tiny charged oil droplets. He found that all charges were integer multiples of a fundamental value, C. Combining this with Thomson’s ratio gave the electron mass: kg.
Models of the Atom
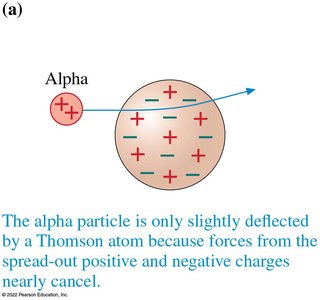
Thomson’s Raisin-Cake Model
Thomson proposed that electrons were embedded in a sphere of positive charge, like raisins in a cake. This model was soon replaced by more accurate models.

Discovery of Radioactivity
Henri Becquerel discovered that uranium emits penetrating rays without an external energy source. Marie and Pierre Curie found other radioactive elements. Three types of radiation were identified:
Alpha rays: Positively charged, stopped by paper, later identified as He2+ ions.
Beta rays: Negatively charged, penetrate metal, identified as high-speed electrons.
Gamma rays: Neutral, highly penetrating, a form of electromagnetic radiation.
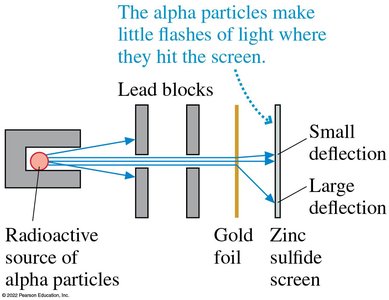
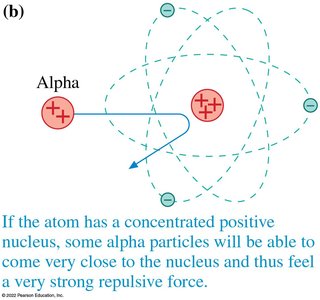
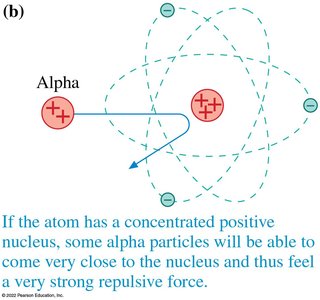
Rutherford’s Gold Foil Experiment
Rutherford showed that most alpha particles passed through gold foil with little deflection, but a few were deflected at large angles. This led to the nuclear model of the atom: a tiny, dense, positively charged nucleus surrounded by electrons.
Discovery of the Neutron
In 1932, the neutron was discovered, completing the basic picture of atomic structure: a nucleus of protons and neutrons surrounded by electrons.
Unanswered Questions and the Need for Quantum Mechanics
By the early 20th century, classical physics could not explain:
Why atoms have discrete spectra
The blackbody spectrum
The cause of radioactivity
The stability of atoms
What holds the atomic nucleus together
These questions led to the development of quantum mechanics, which revolutionized our understanding of matter and energy.
