 Back
BackThe Micro/Macro Connection: Statistical Physics and Thermodynamics
Study Guide - Smart Notes

The Micro/Macro Connection
Introduction to the Micro/Macro Connection
The micro/macro connection in physics explains how the macroscopic properties of matter—such as pressure, temperature, and entropy—arise from the microscopic behavior of atoms and molecules. By understanding the motion and interactions of individual particles, we can predict and explain the observable properties of gases, solids, and liquids.
Macroscopic properties: Pressure (p), temperature (T), volume (V), specific heat, etc.
Microscopic properties: Position, velocity, acceleration, and energy of atoms and molecules.
Key questions addressed:
How do atomic motions determine macroscopic state variables?
How is heat transferred between objects?
Why are most macroscopic processes irreversible?
Molecular Speeds, Collisions, and Mean Free Path
Molecular Motion and Speed Distribution
Gases consist of a vast number of molecules moving randomly and colliding frequently. The distribution of molecular speeds in a gas is not uniform; instead, it follows a statistical pattern, with most molecules having speeds near a certain value (the most probable speed).
Root-mean-square (rms) speed: The square root of the average of the squares of molecular speeds, denoted as .
Distribution: At room temperature, nitrogen molecules have a range of speeds, with a significant fraction between 600 m/s and 700 m/s.

Mean Free Path
The mean free path () is the average distance a molecule travels between collisions. It depends on the number density of molecules and their effective size.
Formula: N/Vr$ is the molecular radius.
For nitrogen at room temperature and 1 atm, nm.

Pressure and Temperature in Gases
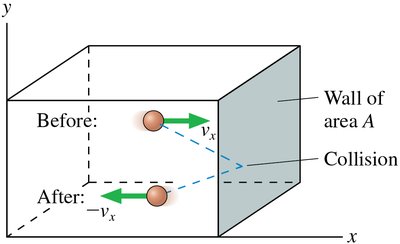
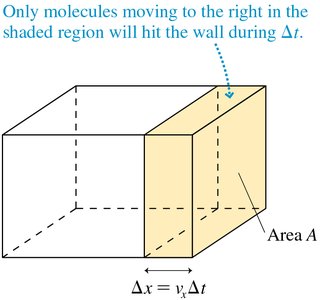
Origin of Gas Pressure
Gas pressure arises from the collisions of molecules with the walls of their container. Each collision imparts a small force, and the cumulative effect of countless collisions produces a measurable pressure.
Pressure formula: NVmv_{\mathrm{rms}}$ is the root-mean-square speed.
Temperature and Kinetic Energy
Temperature is a measure of the average translational kinetic energy of the molecules in a gas. At absolute zero, all molecular motion ceases.
Average kinetic energy per molecule: k_BT$ is the temperature in kelvin.
Root-mean-square speed: $
Thermal Energy and Specific Heat
Thermal Energy in Gases and Solids
The total thermal energy of a system is the sum of the microscopic kinetic and potential energies of its particles. For an ideal monatomic gas, only translational kinetic energy contributes.
Monatomic gas: nR$ is the gas constant.
Change in thermal energy: C_V = \frac{3}{2} R$.

Equipartition Theorem and Degrees of Freedom
The equipartition theorem states that energy is equally distributed among all degrees of freedom (independent modes of energy storage) of a system.
Energy per degree of freedom: $
Degrees of freedom:
Monatomic gas: 3 (translational)
Diatomic gas: 5 (3 translational + 2 rotational)
Elemental solid: 6 (3 translational + 3 vibrational)


Table: Kinetic Theory Predictions
System | Degrees of Freedom | Thermal Energy | Molar Specific Heat |
|---|---|---|---|
Monatomic gas | 3 | ||
Diatomic gas | 5 | ||
Elemental solid | 6 |
Thermal Interactions and Equilibrium
Heat Transfer and Thermal Equilibrium
When two systems at different temperatures are brought into thermal contact, energy is transferred from the hotter to the colder system via collisions at the boundary. This process continues until both systems reach the same average energy per particle, i.e., thermal equilibrium.
Heat (): Energy transferred due to temperature difference.
Thermal equilibrium condition:
Conservation of energy:


Irreversibility, Entropy, and the Second Law
Irreversible Processes and Entropy
Most macroscopic processes are irreversible; for example, heat flows spontaneously from hot to cold, never the reverse. Entropy is a state variable that quantifies the degree of disorder or the number of microscopic configurations (microstates) corresponding to a macroscopic state (macrostate).
Entropy (): \Omega$ is the multiplicity (number of microstates).
Second Law of Thermodynamics:
Formal: The entropy of an isolated system never decreases; it increases until equilibrium is reached.
Informal: Heat flows spontaneously from hot to cold; entropy increases define the arrow of time.
Microstates, Multiplicity, and Probability
Each possible arrangement of particles (microstate) is equally likely, but some macrostates (e.g., equal distribution of energy) are vastly more probable due to their higher multiplicity. The equilibrium state is the most probable macrostate.
Macrostate (Number of Heads) | Multiplicity () |
|---|---|
0 | 1 |
1 | 4 |
2 | 6 |
3 | 4 |
4 | 1 |
Entropy in Physical Processes
Isothermal process: $
Heating at constant pressure: $
Entropy change for an ideal gas: $
Summary of Key Equations and Concepts
Pressure in a gas:
Average kinetic energy:
Thermal energy (monatomic gas):
Entropy:
Second Law: Entropy of an isolated system never decreases.
Applications
Root-mean-square speed: $
Molar specific heats:
Monatomic gas:
Diatomic gas:
Elemental solid:
