 Back
BackThermal Properties of Matter, Calorimetry, and Simple Harmonic Motion
Study Guide - Smart Notes

Thermal Properties of Matter
Specific Heat and Heats of Transformation
When a substance absorbs or releases heat, its temperature may change, or it may undergo a phase change. The amount of heat required depends on the material's specific heat and the heat of transformation for phase changes.
Specific Heat (c): The amount of heat required to raise the temperature of 1 kg of a substance by 1 K (or 1 °C). It is a measure of how resistant a substance is to temperature change.
Heat of Transformation (L): The amount of heat required to change the phase of 1 kg of a substance (e.g., melting or boiling) without changing its temperature.
Key Equations:
For temperature change:
For phase change:
Example: Heating water from 10°C to 100°C and then boiling it involves both temperature change and phase change.
Energy Flow Diagrams and Bar Charts
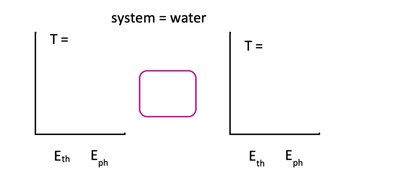
Energy flow diagrams help visualize heat transfer processes. The system (e.g., water) is represented by a box, with arrows indicating heat flow. Energy bar charts show thermal energy (Eth) and phase energy (Eph) before and after heat transfer.
Thermal Energy (Eth): Related to the kinetic energy of particles and proportional to temperature.
Phase Energy (Eph): Related to the arrangement and bonding of particles; increases with less order (gas > liquid > solid).
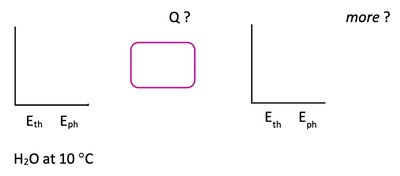
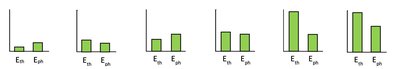
Temperature vs. Heat Transfer Graphs
Graphs of temperature versus heat transfer (Q) show regions of temperature change and regions of phase change. During phase change, temperature remains constant while heat is absorbed or released.

Calorimetry and Thermal Equilibrium
Calorimetry Principles
Calorimetry is the quantitative measurement of heat transfer between substances in thermal contact until equilibrium is reached. The heat lost by one substance equals the heat gained by another (energy conservation).
Conservation of Energy:
Example: A hot metal dropped into cooler water; both reach the same final temperature.

Calorimetry with Phase Change
When a phase change occurs (e.g., ice melting in water), multiple heat transfer processes must be considered: warming the ice, melting it, and warming the resulting water.
Heat Transfer Steps:
Heating ice to 0°C:
Melting ice:
Heating melted water:
Thermal Expansion
Linear and Volumetric Expansion
When a substance changes temperature, its dimensions change due to thermal expansion. The change depends on the material's expansion coefficients.
Linear Expansion:
Volumetric Expansion:
Coefficients: and (units: K-1)

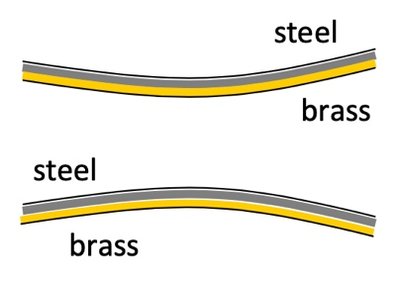
Applications: Bi-metallic Strips and Galileo Thermometers
Bi-metallic strips bend when heated due to different expansion rates of metals. Galileo thermometers use floating bulbs to indicate temperature based on density changes from thermal expansion.

Heat Transfer Mechanisms
Conduction
Conduction is the transfer of thermal energy through a material. The rate depends on the material's thermal conductivity (k), area (A), thickness (L), and temperature difference ().
Equation:
Materials with high k (e.g., metals) conduct heat faster than those with low k (e.g., wood, Styrofoam).

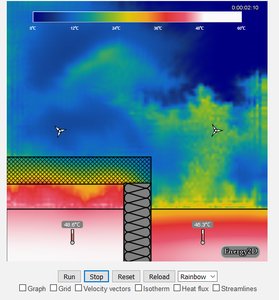
Convection
Convection is the transfer of heat by the movement of fluids (liquids or gases). It occurs in open systems and is influenced by temperature differences and fluid motion.
Radiation
Radiation is the transfer of energy by electromagnetic waves, such as infrared radiation. All objects emit radiation depending on their temperature and emissivity.
Stefan-Boltzmann Law:
= emissivity, = Stefan-Boltzmann constant ( W/m2K4), = area, = temperature in K

Simple Harmonic Motion (SHM)
Characteristics of SHM
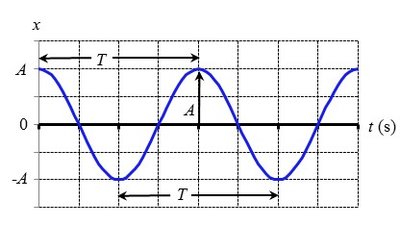
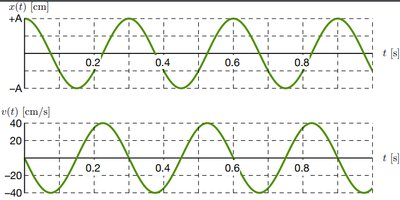
Simple harmonic motion describes periodic oscillations, such as a mass on a spring or a pendulum. The motion is characterized by amplitude (A), period (T), and frequency (f).
Position as a function of time:
Frequency:
Velocity:
Mass-Spring System and Hooke's Law
The restoring force in a mass-spring system is described by Hooke's Law:
Hooke's Law:
Period of oscillation:
Stiffer springs (higher k) result in shorter periods.
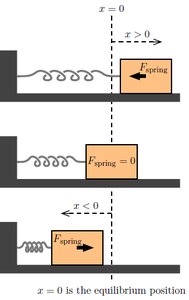
Energy in SHM
Energy in SHM is conserved and alternates between kinetic energy (KE) and potential energy (PE) stored in the spring.
Kinetic Energy:
Spring Potential Energy:
Total Energy: (constant)
Waves and Superposition
Wave Pulses and Reflection
Waves are disturbances that travel through a medium. They can be longitudinal (disturbance parallel to direction) or transverse (disturbance perpendicular).
Reflection at Fixed End: Pulse inverts upon reflection.
Reflection at Free End: Pulse reflects without inversion.
Tables
Table: Selected Specific Heats, Melting and Boiling Points, and Heats of Transformation
Substance | c (solid) (J/kg·K) | c (liquid) (J/kg·K) | Tmelt (°C) | Lf (J/kg) | Tboil (°C) | Lv (J/kg) |
|---|---|---|---|---|---|---|
Water | 2090 | 4190 | 0 | 3.33 × 105 | 100 | 22.6 × 105 |
Ethyl alcohol | - | 2400 | -114 | 1.09 × 105 | 78 | 8.79 × 105 |
Mercury | - | 140 | -39 | 0.11 × 105 | 357 | 2.96 × 105 |
Lead | 128 | - | 328 | 0.25 × 105 | 1750 | 8.58 × 105 |
Aluminum | 900 | - | - | - | - | - |
Copper | 385 | - | - | - | - | - |
Table: Thermal Expansion Coefficients
Substance | Linear: α (K-1) | Volume: β (K-1) |
|---|---|---|
Water | - | 210 × 10-6 |
Alcohol | - | 1100 × 10-6 |
Kerosene | - | 990 × 10-6 |
Glass | 9 × 10-6 | 27 × 10-6 |
Steel | 12 × 10-6 | 36 × 10-6 |
Concrete | 12 × 10-6 | 36 × 10-6 |
Brass | 19 × 10-6 | 57 × 10-6 |
Table: Thermal Conductivity Values
Material | k (W/m·K) |
|---|---|
Copper | 400 |
Aluminum | 205 |
Stainless steel | 14 |
Ice | 1.7 |
Glass | 1.05 |
Skin | 0.50 |
Wood | 0.2 |
Paper | 0.05 |
Styrofoam | 0.033 |
Air | 0.026 |
Table: Emissivity Values
Substance/Material | Emissivity, e |
|---|---|
Skin | 0.95 – 0.98 |
Wood | 0.9 |
Polished aluminum | 0.05 |
Brick | 0.8 – 0.9 |
Styrofoam | 0.6 |
Additional info: These notes cover the core concepts of thermal properties of matter, calorimetry, thermal expansion, heat transfer mechanisms, and simple harmonic motion, with relevant equations, diagrams, and tables for college-level physics.
