 Back
BackThermodynamics: Heat Capacities, Heat Engines, and the Laws of Thermodynamics- lecture 30
Study Guide - Smart Notes

Thermodynamics
Specific Heat Capacities
Specific heat capacity is a fundamental property that quantifies the amount of heat required to change the temperature of a substance. In thermodynamics, it is often useful to express heat in terms of moles for ideal gases.
Molar Specific Heat Capacity (C): The heat required to raise the temperature of one mole of a substance by one Kelvin, measured in J/(mol·K).
Heat Equation:
Monatomic Ideal Gas:
At constant pressure:
At constant volume:
Specific heat ratio:
Difference:
Adiabatic Expansion & Compression
Adiabatic processes occur without heat exchange with the environment. The relationship between pressure and volume for an ideal gas undergoing adiabatic expansion or compression is governed by the specific heat ratio.
Adiabatic Equation:
Specific Heat Ratio (): Depends on the gas type (e.g., for monatomic gases).
Example: For a monatomic ideal gas with Pa, m3, Pa:
Result: m3
Heat Engines
The Second Law of Thermodynamics
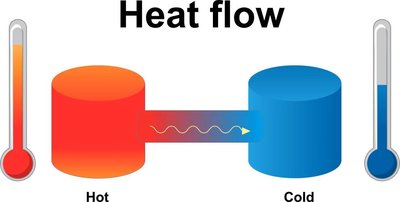
The second law states that heat naturally flows from hotter to colder material, and this process is irreversible. Heat cannot spontaneously flow from cold to hot.
Irreversibility: Natural heat flow is always from hot to cold.
Heat Engine Fundamentals
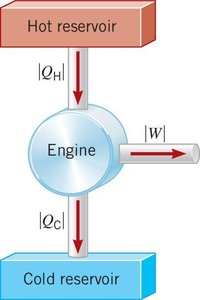
A heat engine is a device that converts heat energy into mechanical work by operating between two thermal reservoirs.
Three Fundamental Processes:
Heat is supplied from the hot reservoir.
Engine uses part of the heat to perform work.
Remaining heat flows to the cold reservoir.
Efficiency of a Heat Engine
Efficiency measures how effectively a heat engine converts input heat into work. It is always less than 100% due to unavoidable heat loss.
Efficiency Equation:
Energy Conservation:
Work Done:
Efficiency in terms of waste heat:
Real Engines: Always have some heat loss (), so .
Carnot Engine
The Carnot engine is a theoretical model for the most efficient heat engine possible, operating via reversible processes between two reservoirs.
Carnot Principle: No engine operating between two reservoirs can be more efficient than a Carnot engine.
Efficiency depends only on reservoir temperatures:
Heat ratio:
Carnot efficiency:
Example: For K, K:
(6.036%)
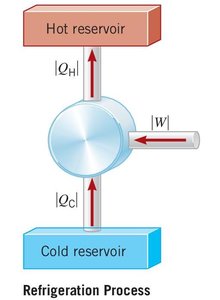
Reverse Heat Engine (Refrigeration Process)
Reverse heat engines, such as refrigerators and air conditioners, use work to transfer heat from a cold reservoir to a hot reservoir.
Coefficient of Performance (COP):
Refrigerator/air conditioner:
Heat pump:
Typical values: 2–6 for refrigerators/air conditioners, 3–4 for heat pumps.
Entropy
Definition and Properties
Entropy is a measure of disorder or randomness in a system. It increases in irreversible processes and remains constant in reversible processes.
Change in Entropy:
Isolated System: Total entropy cannot decrease; it increases or stays constant.
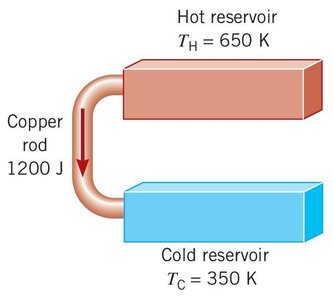
Entropy Example
When 1200 J of heat flows irreversibly from a hot reservoir ( K) to a cold reservoir ( K), the change in entropy of the Universe can be calculated.
Heat leaving hot reservoir: J/K
Heat entering cold reservoir: J/K
Total change: J/K
The Laws of Thermodynamics
Summary of the Laws
The four fundamental laws govern all thermodynamic processes:
Zeroth Law: If two systems are each in thermal equilibrium with a third, they are in thermal equilibrium with each other.
First Law: Conservation of energy:
Second Law: Natural processes are irreversible; entropy of an isolated system increases or remains constant.
Third Law: Absolute zero ($0$ K) is unattainable; reaching it would require infinite energy.
Absolute Zero and the Third Law
Absolute zero is the lowest possible temperature, where the internal energy of matter is minimized. The third law states that it is impossible to reach absolute zero.
Absolute Zero: $0= -273.15= -459.67$ °F
Zero-point energy: The lowest possible internal energy.
Impossibility: Infinite energy required to reach absolute zero.
