 Back
BackThermodynamics, Ideal Gas Law, and Kinetic Theory: Study Notes- lecture 29
Study Guide - Smart Notes

The Ideal Gas Law and Avogadro's Number
Avogadro's Number and the Mole
The mole (mol) is the SI unit for measuring the amount of substance, defined as the number of atoms in 12 grams of carbon-12. This number is known as Avogadro's Number:
Avogadro's Number (N_A): atoms/mol
Number of moles () in a sample: , where is the number of particles.
Alternatively, , where is the sample mass and is the molar mass.

Additional info: The image humorously represents Avogadro's Number by writing it on an avocado, reinforcing the concept for students.
The Ideal Gas Law
The Ideal Gas Law describes the relationship between pressure, volume, temperature, and the number of moles in a gas:
is the universal gas constant:
Alternate form: , where is the number of particles and is the Boltzmann constant ()
The law is derived from Boyle's, Charles', Avogadro's, and Gay-Lussac's Laws.
Example: Volume Change with Temperature
When mass and pressure are constant, the ratio of volume to temperature remains constant:
Final volume:
Example: If , , , then
Kinetic Theory of Gases
Distribution of Speeds
In a gas, particles move at a range of speeds. The distribution of these speeds depends on temperature:
Higher temperature increases both the most probable speed and the spread of speeds.
Statistical methods are used to describe the behavior of large numbers of particles.
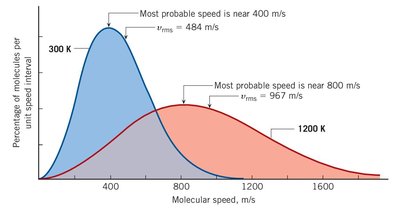
Additional info: The image shows the Maxwell speed distribution for 300 K and 1200 K, illustrating how temperature affects molecular speed.
Root-Mean-Square Speed
The root-mean-square speed () is a statistical measure of the speed of particles in a gas:
is the average of the squared speeds.
Note:
Monatomic Ideal Gas
For a monatomic ideal gas (single atoms, not molecules):
Three degrees of freedom (motion in x, y, z directions)
Average kinetic energy:
Internal energy:
Thermodynamics
Basic Concepts
Thermodynamics is the study of heat, work, and energy transfer. A system is the collection of objects under study, and the environment is everything else. Energy transfer from the environment to the system is considered positive in physics.
Walls: Diathermal and Adiabatic
Systems are separated from their environment by walls:
Diathermal walls: Allow heat to flow between systems.
Adiabatic walls: Do not allow heat flow; systems are insulated.
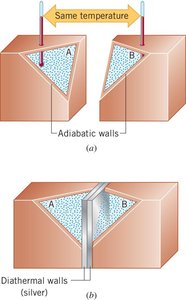
Additional info: The image illustrates the difference between adiabatic and diathermal walls, showing how heat transfer is blocked or allowed.
The Zeroth Law of Thermodynamics
The zeroth law establishes the concept of thermal equilibrium:
If two objects are at the same temperature, no net heat flows between them.
If A is in equilibrium with C, and C with B, then A is in equilibrium with B.
Thermal Processes
The First Law of Thermodynamics
The first law relates internal energy change to heat and work:
Work is positive when done by the system, negative when done on the system.
Types of Thermal Processes
Four main quasi-static processes are considered:
Isobaric: Constant pressure (; )
Isochoric: Constant volume (; )
Isothermal: Constant temperature (; ; )
Adiabatic: No heat transfer (; ; )
Complex Processes and PV Graphs
For processes not fitting the four main types, the work done can be found from the area under a pressure-volume (PV) graph:
Work done = Area under the PV curve
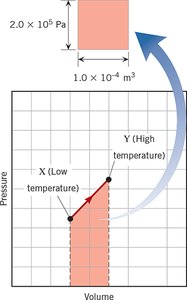
Additional info: The image shows a PV graph, illustrating how work is calculated as the area under the curve.
Thermodynamics Example: Energy Replacement
Example calculation for energy lost during exercise:
Power output:
Time:
Work done:
Heat lost:
Internal energy lost:
Energy per banana:
Bananas needed: bananas
Process | Constant | Work Done | Internal Energy Change |
|---|---|---|---|
Isobaric | Pressure | ||
Isochoric | Volume | ||
Isothermal | Temperature | ||
Adiabatic | No heat transfer |
Additional info: Table summarizes the main thermal processes, their constants, work done, and internal energy changes.
