 Back
BackThermodynamics II: Kinetic Theory, Specific Heat, and Heat Transfer
Study Guide - Smart Notes

Thermodynamics II: Kinetic Theory, Specific Heat, and Heat Transfer
Microscopic (Statistical) Approach to Thermodynamics
Thermodynamics can be explained using Newtonian physics by considering the microscopic behavior of particles. The kinetic theory provides a statistical explanation for macroscopic phenomena such as pressure, temperature, and internal energy by relating them to the positions, speeds, and energies of constituent particles.
Kinetic Theory: Describes gases as large numbers of small particles (atoms or molecules) in constant, random motion.
Macroscopic Quantities: Pressure (P), Temperature (T), Internal Energy (U), and Entropy (S) are derived from microscopic properties.
The Ideal Gas Law and Boltzmann Constant
The ideal gas law relates the pressure, volume, and temperature of an ideal gas. It can be expressed in terms of the number of molecules and the Boltzmann constant:
Equation:
Boltzmann Constant:
Microscopic Form:

Kinetic Energy and Molecular Speeds
The average kinetic energy of a gas molecule is directly proportional to the temperature. The root-mean-square (rms) speed is a useful measure of the average speed of gas molecules.
Average Kinetic Energy:
Root-Mean-Square Speed:
Example: For at room temperature,
Mean Free Path
The mean free path is the average distance a molecule travels between collisions. It depends on temperature, pressure, and molecular diameter.
Equation:
Variables: = temperature, = pressure, = molecular diameter
Example: For at 1 atm,
Concepts: Temperature, Thermal Energy, Internal Energy, and Heat
It is important to distinguish between temperature, thermal energy, internal energy, and heat:
Temperature (T): Measures the average kinetic energy of particles; indicates how hot or cold an object is.
Thermal Energy (E_{th}): Total kinetic energy of all molecules (includes translational, rotational, and vibrational).
Internal Energy (U): Total energy of a system, including kinetic and potential energies.
Heat (Q): Energy transferred due to temperature difference.
Specific Heat and Heat Capacity
Specific heat is the amount of heat required to raise the temperature of a unit mass of a substance by 1°C (or 1 K). Heat capacity is the amount of heat required to raise the temperature of an object by 1°C.
Specific Heat (c):
Heat Capacity (C):
Units: J/kg·°C or cal/g·°C
1 calorie (cal):
1 Calorie (food):
Heat Transfer and Temperature Change
When heat is added to a substance, its temperature rises, kinetic energy increases, and expansion may occur. If enough heat is added, a phase transition can take place (solid → liquid → gas).
Equation:
Sign Convention: (heat added), (heat removed)
Thermal Equilibrium
When two objects of different temperatures are placed in thermal contact, heat flows from the hotter to the cooler object until thermal equilibrium is reached (final temperatures are equal).
Example: Two blocks of the same material, one with mass at C and one with mass at C, reach equilibrium at .
Calculation: Set and solve for :
C$ 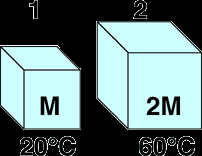
Worked Examples
Example 1: How much heat is needed to raise 1.0 g of water from 14.5°C to 15.5°C? J/g·°C
J
Example 2: Heating water in an aluminum pan: 0.500 kg aluminum pan heats 0.250 kg water from 20.0°C to 80.0°C. J/kg·°C, J/kg·°C
J J J
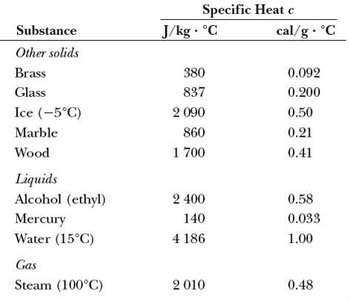
Table: Specific Heats of Common Substances
The following tables summarize the specific heats of various solids, liquids, and gases. These values are essential for solving heat transfer problems.
Substance | J/kg·°C | cal/g·°C |
|---|---|---|
Aluminum | 900 | 0.215 |
Beryllium | 1830 | 0.436 |
Cadmium | 230 | 0.055 |
Copper | 387 | 0.0924 |
Germanium | 322 | 0.077 |
Gold | 129 | 0.0308 |
Iron | 448 | 0.107 |
Lead | 128 | 0.0305 |
Silicon | 703 | 0.168 |
Silver | 234 | 0.056 |
Substance | J/kg·°C | cal/g·°C |
|---|---|---|
Brass | 380 | 0.092 |
Glass | 837 | 0.200 |
Ice (-5°C) | 2090 | 0.50 |
Marble | 860 | 0.21 |
Wood | 1700 | 0.41 |
Alcohol (ethyl) | 2400 | 0.58 |
Mercury | 140 | 0.033 |
Water (15°C) | 4186 | 1.00 |
Steam (100°C) | 2010 | 0.48 |
Summary Table: Key Thermodynamic Quantities
Quantity | Symbol | Definition | SI Unit |
|---|---|---|---|
Temperature | T | Average kinetic energy of particles | K (Kelvin) |
Thermal Energy | E_{th} | Total kinetic energy of all molecules | J (Joule) |
Internal Energy | U | Total energy (kinetic + potential) | J (Joule) |
Heat | Q | Energy transferred due to temperature difference | J (Joule) |
Specific Heat | c | Heat required to raise 1 kg by 1°C | J/kg·°C |
