 Back
BackThermodynamics: Temperature, Heat, and the First Law
Study Guide - Smart Notes

Thermodynamics: Temperature, Heat, and the First Law
The Zeroth Law of Thermodynamics
The zeroth law of thermodynamics establishes the concept of temperature and thermal equilibrium. It states that if two bodies (A and B) are each in thermal equilibrium with a third body (T), then A and B are in thermal equilibrium with each other. This principle allows the use of thermometers to measure temperature consistently.
Thermal equilibrium: Two systems are in thermal equilibrium if there is no net flow of energy between them when they are in contact.
Temperature: A measure of the average kinetic energy of the particles in a substance.
Thermal energy: The total internal energy (kinetic + potential) of all particles in a system.
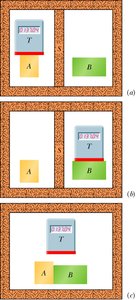
Example: If a thermometer reads the same temperature when in contact with two different objects, those objects are in thermal equilibrium with each other.
Measurement of Temperature and Temperature Scales
Temperature is measured by observing a property of a thermometer in thermal equilibrium with the system. Common temperature scales include Celsius (°C), Fahrenheit (°F), and Kelvin (K). The Kelvin scale is the SI unit and is based on absolute zero, the point where molecular kinetic energy is zero.
Kelvin to Celsius:
Celsius to Fahrenheit:
Absolute zero: 0 K (theoretical minimum temperature)
Example: Room temperature is approximately 290 K (17°C).
Thermal Expansion
When the temperature of a solid or liquid increases, its dimensions expand. For a rod of length L, the change in length due to a temperature change is:
Linear expansion:
Volume expansion:
Where is the coefficient of linear expansion and is the coefficient of volume expansion.
Example: A diesel tanker loses about 2% of its volume when cooled by 23 K, due to thermal contraction.
Heat and Heat Capacity
Heat is the energy transferred between systems due to a temperature difference. The SI unit is the Joule (J), but calories (cal) and British thermal units (Btu) are also used. Heat is not a property possessed by a body, but a process of energy transfer.
Heat capacity (C): The amount of heat required to change a body's temperature by one degree.
Specific heat capacity (c): Heat capacity per unit mass:
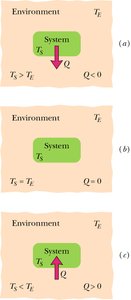
Sign convention: if heat is absorbed by the system; if heat is released.
Example: Calculating the specific heat of a metal by mixing it with water and measuring the equilibrium temperature.
Heats of Transformation (Latent Heat)
When a substance changes phase (e.g., solid to liquid), energy is absorbed or released without a temperature change. The energy per unit mass required is called the latent heat (L):
Heat of fusion (LF): For melting/freezing
Heat of vaporization (LV): For boiling/condensation
Example: Calculating the total heat required to convert ice at -10°C to water at 15°C involves three steps: heating the ice, melting it, and heating the resulting water.
The First Law of Thermodynamics
The first law of thermodynamics is a statement of energy conservation for thermodynamic systems. It relates changes in internal energy () to heat (Q) and work (W):
Adiabatic process:
Constant-volume process:
Adiabatic free expansion:
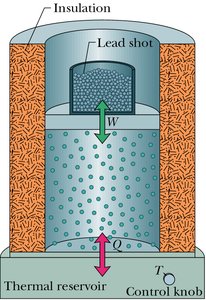
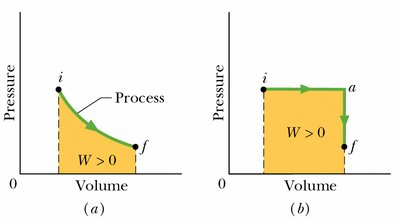
Work done by a gas: (area under the p-V curve)
Example: Calculating the work, heat, and change in internal energy when water boils and turns to steam at constant pressure.
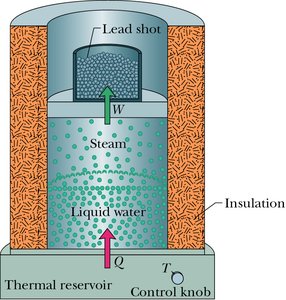
Heat Transfer Mechanisms
There are three main mechanisms for heat transfer:
Conduction: Transfer of energy through collisions between particles in a solid. The rate of heat flow is:
Where k is the thermal conductivity, A is the cross-sectional area, and are the temperatures at each end, and L is the length.
Convection: Transfer of heat by the movement of fluids (liquids or gases) due to temperature differences.
Radiation: Transfer of energy by electromagnetic waves. The rate of radiative energy emission is:
Where is the Stefan-Boltzmann constant and is the emissivity of the surface.
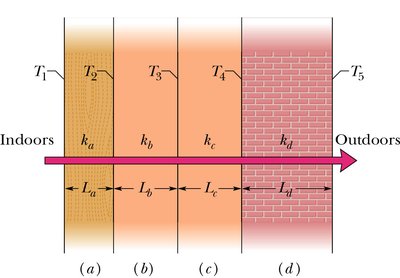
Example: Calculating the interface temperature in a composite wall using steady-state conduction and thermal conductivities of different materials.
Additional info: The notes above include expanded academic context and examples for clarity and completeness, as well as all relevant equations and image explanations.
