 Back
BackWork and Energy in Thermodynamic Processes
Study Guide - Smart Notes

Work in Thermodynamic Processes
Introduction to Thermodynamic Work
In thermodynamics, the work done by or on a system depends on the specific process or path taken between two states. The calculation of work, heat, and change in internal energy (ΔU) varies depending on the type of process. The four principal thermodynamic processes are: isothermal (constant temperature), isochoric (constant volume), isobaric (constant pressure), and adiabatic (no heat exchange).
Isothermal: Temperature remains constant.
Isochoric: Volume remains constant.
Isobaric: Pressure remains constant.
Adiabatic: No heat enters or leaves the system.
For all these processes, the First Law of Thermodynamics applies: .
Calculation of Work, Heat, and Internal Energy
General Approach
To analyze thermodynamic processes, we use:
Equation of the process: Specifies which variable is held constant.
Equation of state: For ideal gases, .
Assume processes are reversible (quasi-static), maintaining thermal equilibrium at each step.
Isochoric Process (Constant Volume)
Key Features
In an isochoric process, the volume does not change (), so the system does no work (). All energy added as heat increases the internal energy.
Work:
Change in Internal Energy:
Example: Heating a gas in a rigid, closed container.
For an ideal gas at constant volume:
Isobaric Process (Constant Pressure)
Key Features
In an isobaric process, pressure remains constant while volume and temperature may change. The work done is proportional to the change in volume.
Work:
Heat: can be calculated using the heat capacity at constant pressure ().
Example: Boiling water at atmospheric pressure.
For an ideal gas at constant pressure:
Isothermal Process (Constant Temperature)
Key Features
In an isothermal process, temperature remains constant, but both pressure and volume change. The work done is given by:
For an ideal gas:
Compression (): is negative (work done on the system).
Expansion (): is positive (system does work on surroundings).
Adiabatic Process (No Heat Exchange)
Key Features
In an adiabatic process, no heat is transferred (). The process can be achieved by thermal insulation or by carrying out the process rapidly. The internal energy change equals the negative of the work done:
Described by: , where
For an ideal gas:
Values of :
Monatomic gases:
Diatomic gases:
Work done in a reversible adiabatic process:
Temperature change in adiabatic process:
Adiabatic compression: increases
Adiabatic expansion: decreases
Examples of Adiabatic Processes
Thermally insulated system: e.g., Dewar flask prevents heat flow.
Rapid expansion: e.g., popping a champagne cork—expansion is so fast that little heat is exchanged, resulting in a temperature drop and condensation of water vapor.


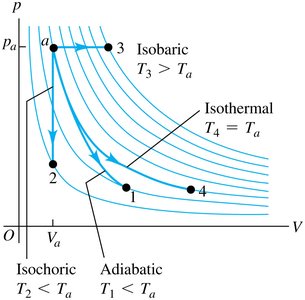
Comparing Thermodynamic Processes on a pV Diagram
pV Diagram Representation
The four processes—isochoric, isobaric, isothermal, and adiabatic—can be represented on a pressure-volume (pV) diagram. Each process has a characteristic curve:
Isochoric: Vertical line (constant volume)
Isobaric: Horizontal line (constant pressure)
Isothermal: Hyperbolic curve (constant temperature)
Adiabatic: Steeper curve than isothermal (no heat exchange)
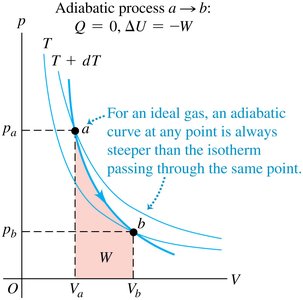
Adiabatic vs. Isothermal Curves
For an ideal gas, the adiabatic curve is always steeper than the isothermal curve passing through the same point. This is because for all real gases.
Summary Table: Thermodynamic Processes
Process | Constant Variable | Work () | Heat () | Internal Energy Change () |
|---|---|---|---|---|
Isochoric | Volume () | $0$ | ||
Isobaric | Pressure () | |||
Isothermal | Temperature () | $0$ | ||
Adiabatic | No heat () | $0$ |
Additional info: The equations and relationships above assume ideal gas behavior and reversible (quasi-static) processes. Real gases and irreversible processes may deviate from these results.
