 Back
BackX-Rays, X-Ray Diffraction, and the Photoelectric Effect
Study Guide - Smart Notes

X-Rays and X-Ray Diffraction
X-Ray Imaging and Computed Tomography (CT/CAT Scans)
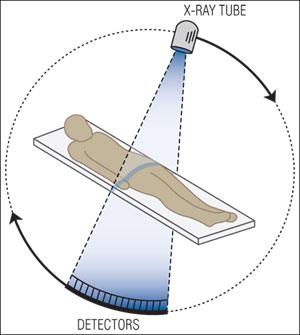
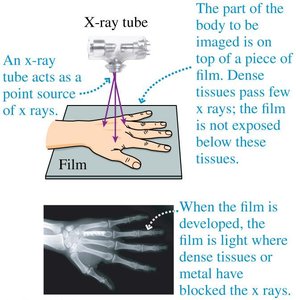
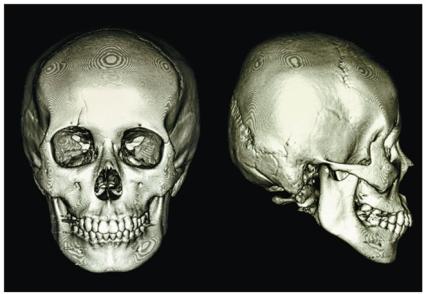
X-rays are a form of high-energy electromagnetic radiation used extensively in medical imaging. When x-rays pass through the body, dense tissues such as bone absorb more x-rays and appear lighter on the resulting image, while less dense tissues allow more x-rays to pass through and appear darker. Computed Axial Tomography (CAT or CT) scans use rotating x-ray beams and detectors to create cross-sectional or 3D images of the body, providing detailed internal views for diagnostic purposes.
X-ray images are produced by differential absorption of x-rays by various tissues.
CT scans use multiple x-ray images taken from different angles to reconstruct a 3D image.
Applications include medical diagnostics, security scanning, and material analysis.
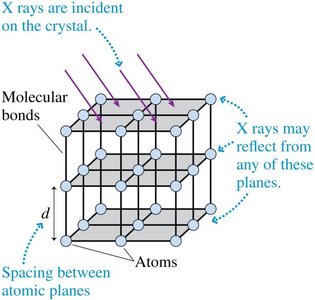
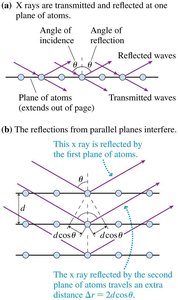
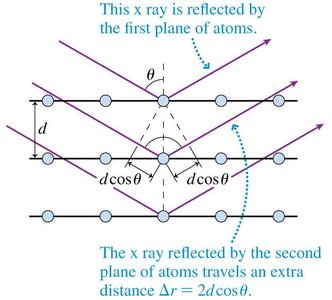
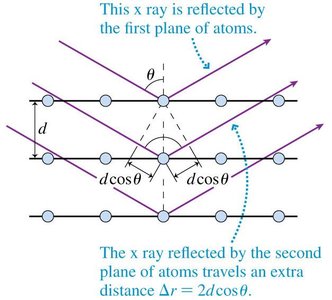
X-Ray Diffraction and the Bragg Condition
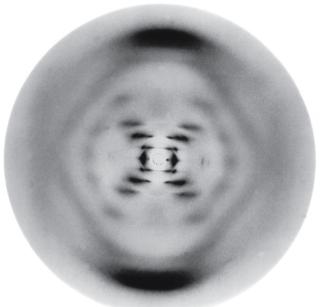
X-ray diffraction is a powerful technique for studying the atomic structure of crystals. When x-rays are incident on a crystalline material, they are scattered by the regularly spaced planes of atoms. Constructive interference occurs at specific angles, producing a diffraction pattern that reveals information about the crystal structure. The condition for constructive interference is given by the Bragg equation:
Bragg Condition: , where is the spacing between atomic planes, is the angle of incidence for the -th order maximum, is the x-ray wavelength, and $m$ is an integer (order of maximum).
X-ray diffraction patterns are used to determine the arrangement of atoms in crystals, such as the famous "Photo 51" image of DNA.
The Photoelectric Effect
Characteristics and Experimental Setup
The photoelectric effect is the emission of electrons from a material (typically a metal) when it is exposed to light of sufficient frequency. This phenomenon provided crucial evidence for the quantum nature of light. The basic experimental setup involves shining light onto a metal surface and measuring the resulting current as electrons are ejected.
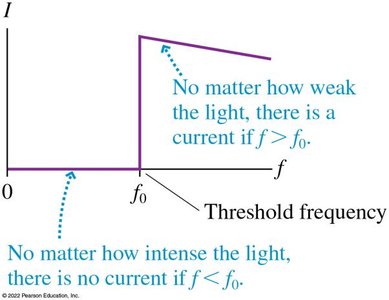
Electrons are only emitted if the light frequency exceeds a threshold frequency .
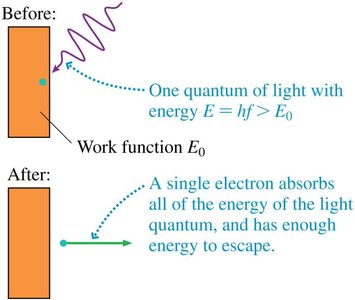
The work function is the minimum energy required to remove an electron from the metal.
The maximum kinetic energy of emitted electrons is .
The stopping potential is related to the maximum kinetic energy by .
Key Observations and Einstein's Explanation
Increasing light intensity increases the number of emitted electrons but not their maximum energy.
Increasing the frequency of light (above ) increases the maximum kinetic energy of electrons.
No electrons are emitted if the frequency is below , regardless of intensity.
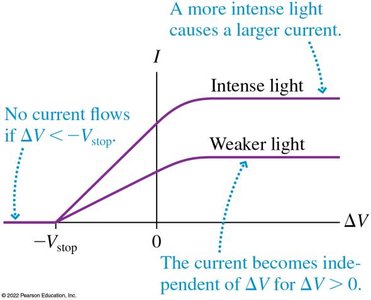
Einstein explained the photoelectric effect by proposing that light consists of discrete packets of energy called photons. Each photon has energy , where is Planck's constant and is the frequency of light. An electron absorbs a single photon; if the photon's energy exceeds the work function, the electron is emitted with kinetic energy .
Applications and Examples
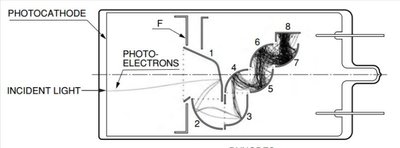
Photoelectric effect apparatus is used to measure Planck's constant and study quantum phenomena.
Photomultiplier tubes and solar cells operate based on the photoelectric effect.



Photons and Wave-Particle Duality
Wave-Particle Duality of Light
Light exhibits both wave-like and particle-like properties, a concept known as wave-particle duality. In the double-slit experiment, even when photons are sent one at a time, an interference pattern emerges over time, demonstrating the wave nature of light. However, each photon is detected as a discrete particle.
Wave-particle duality is a fundamental concept in quantum physics.
Photomultiplier tubes are used to detect individual photons in experiments.
