 Back
BackBiological Macromolecules: Structure, Function, and Types
Study Guide - Smart Notes

Biological Macromolecules
Introduction to Macromolecules
Biological macromolecules are large, complex molecules essential for life. They include carbohydrates, proteins, nucleic acids, and lipids. Except for lipids, these molecules are polymers, built from repeating monomer units through specific chemical reactions.
Polymers: Long chains of monomers linked by covalent bonds.
Macromolecules: Large molecules, typically polymers, with critical biological functions.
Four Major Classes: Carbohydrates, Proteins, Nucleic Acids (all polymers), and Lipids (not true polymers).
Polymer Formation and Breakdown
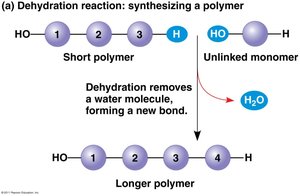
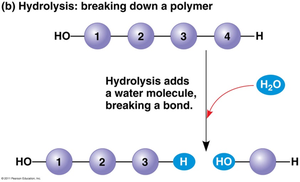
Polymers are synthesized and degraded by two main types of reactions: dehydration synthesis and hydrolysis.
Dehydration Synthesis: Monomers are joined by removing a water molecule, forming a new covalent bond.
Hydrolysis: Polymers are broken down into monomers by the addition of water, breaking the covalent bond.
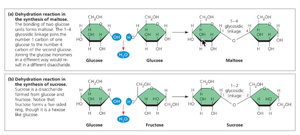
Example: The formation of maltose (a disaccharide) from two glucose molecules involves a dehydration reaction, while its breakdown involves hydrolysis.
Carbohydrates
Structure and Function
Carbohydrates are organic molecules with the general formula (CH2O)n. They serve as energy sources and structural materials in cells.
Monosaccharides: Simple sugars (e.g., glucose, fructose, ribose) that are hydrophilic and serve as monomers for larger carbohydrates.
Disaccharides: Composed of two monosaccharides joined by glycosidic linkages (e.g., maltose, sucrose, lactose).
Polysaccharides: Long chains of monosaccharides; can be storage (starch, glycogen) or structural (cellulose, chitin).
Types of Polysaccharides
Storage Polysaccharides: Starch (plants), Glycogen (animals)
Structural Polysaccharides: Cellulose (plant cell walls), Chitin (exoskeletons of invertebrates)
Monomer Differences: Starch is composed of α-glucose (OH on C1 below the ring), while cellulose is composed of β-glucose (OH on C1 above the ring).
Lipids
Structure and Types
Lipids are hydrophobic molecules that serve as energy stores, structural components of membranes, and signaling molecules. They are not true polymers.
Triglycerides: Composed of glycerol and three fatty acids; can be saturated (no double bonds) or unsaturated (one or more double bonds).
Phospholipids: Contain glycerol, two fatty acids, and a phosphate group; amphiphilic, forming the basis of cell membranes.
Steroids: Four-ring structures; cholesterol is the precursor for steroid hormones.
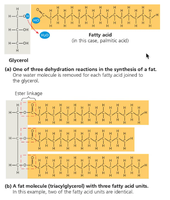
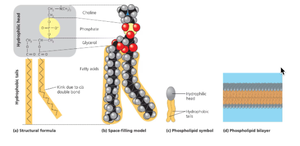
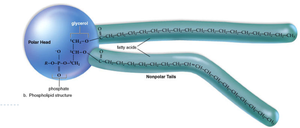
Proteins
Structure and Function
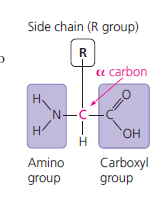
Proteins are polymers of amino acids, folded into specific three-dimensional shapes that determine their function. They perform a vast array of cellular tasks.
Monomer: Amino acid (20 types, differing in R group/side chain)
Peptide Bond: Covalent bond joining amino acids via dehydration synthesis
Functions: Enzyme catalysis, defense, transport, support, motion, regulation, storage
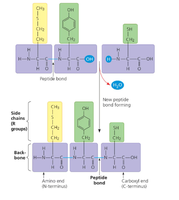
Levels of Protein Structure
Primary Structure: Linear sequence of amino acids
Secondary Structure: Local folding (α-helix, β-sheet) stabilized by hydrogen bonds
Tertiary Structure: Overall 3D shape, stabilized by various bonds and interactions
Quaternary Structure: Association of multiple polypeptide chains (in some proteins)
Types of Bonds: Peptide (primary), hydrogen (secondary), disulfide, ionic, hydrophobic interactions (tertiary and quaternary)
Protein Functions and Examples
Enzymes: Catalyze biochemical reactions
Structural Proteins: Collagen (fibrous, hydrophobic)
Functional Proteins: Hemoglobin (globular, hydrophilic)
Denaturation: Loss of structure and function due to heat or pH changes
Nucleic Acids
Structure and Function
Nucleic acids are polymers of nucleotides, responsible for storing and transmitting genetic information. Types include DNA, RNA, and ATP.
Monomer: Nucleotide (phosphate group, sugar, nitrogenous base)
DNA: Double helix, deoxyribose sugar, bases A, T, C, G; stores genetic information
RNA: Single-stranded, ribose sugar, bases A, U, C, G; involved in gene expression
ATP: Adenosine triphosphate, energy currency of the cell
Base Pairing: A pairs with T (or U in RNA), C pairs with G via hydrogen bonds.
Summary Table: Major Biological Macromolecules
Class | Monomer | Bond Type | Function |
|---|---|---|---|
Carbohydrates | Monosaccharide | Glycosidic linkage | Energy, structure |
Lipids | Fatty acids, glycerol | Ester linkage | Energy storage, membranes |
Proteins | Amino acid | Peptide bond | Catalysis, structure, transport |
Nucleic Acids | Nucleotide | Phosphodiester bond | Genetic information |
Additional info: This summary covers the structure, synthesis, and function of the four major classes of biological macromolecules, as outlined in introductory college biology courses (Ch. 5: The Structure and Function of Large Biological Molecules).
